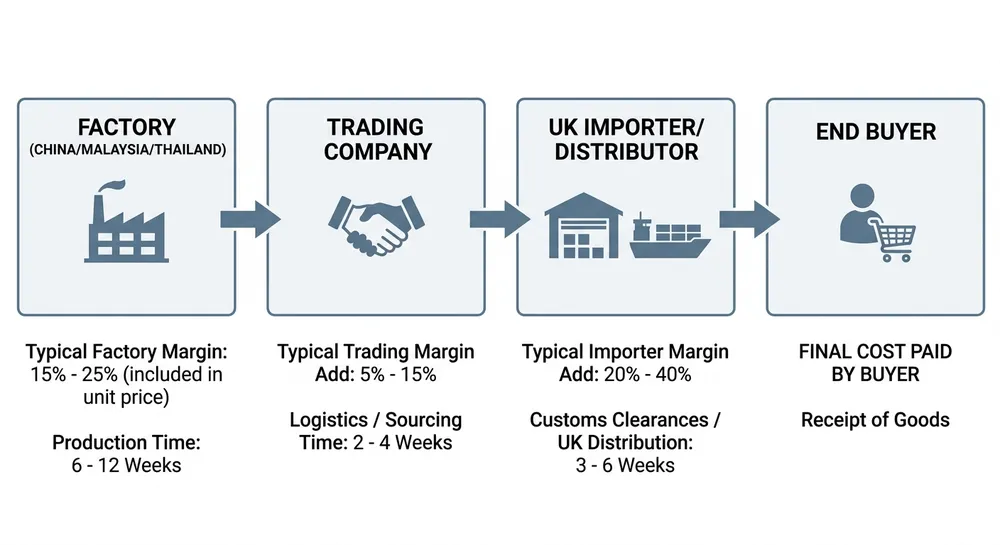
The UK market for blue nitrile gloves runs through a layered supply chain that most importers never fully see. At the front end, you have domestic distributors and PPE wholesalers offering next-day delivery and familiar account terms. Behind them — usually two or three steps removed — are the factories in China, Malaysia, and Thailand that actually make the product. That distance between buyer and manufacturer is where pricing opacity, spec inconsistency, and compliance uncertainty tend to accumulate.
This article maps the current supplier landscape for UK importers sourcing blue nitrile gloves at container scale. It covers what EN 455 compliance actually requires from your supplier's documentation, how post-Brexit UKCA/CE marking affects your import paperwork, and what separates a factory worth dealing with directly from one that will cost you a rejected container at Felixstowe.
The 10 suppliers profiled below represent the realistic range of options available to a UK importer today — from domestic PPE distributors with strong local logistics to factory-direct manufacturers with the certifications and capacity to supply at volume. The goal is not a ranked list. It is a sourcing framework that helps you decide which model fits your procurement situation.
Why UK Importers Struggle to Find Factory-Direct Blue Nitrile Glove Suppliers
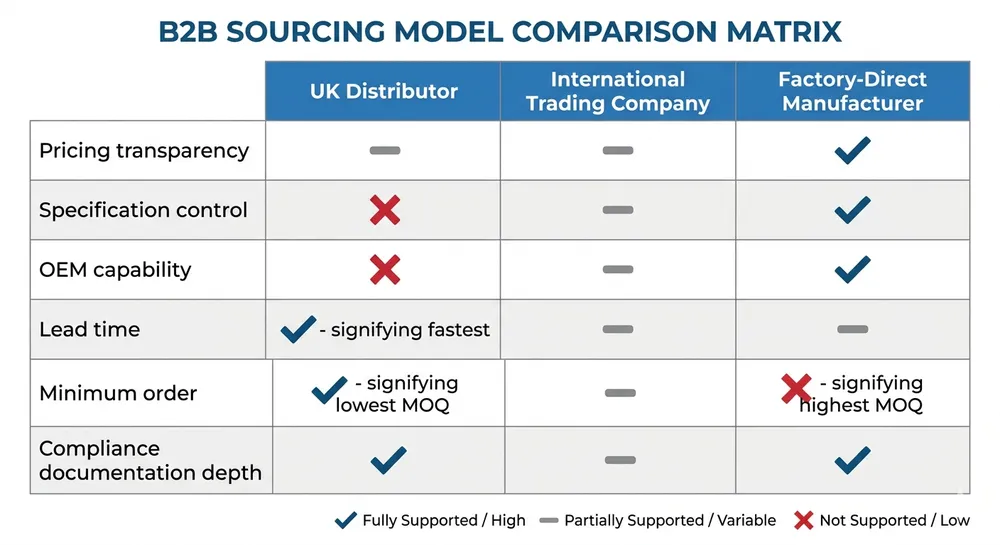
The structural problem is not a shortage of suppliers — it is a shortage of visibility. Search for "blue nitrile gloves suppliers UK" and you will find pages of distributors, PPE wholesalers, and aggregator listings. Most of them are buying from the same three or four factories in Shandong or Selangor and reselling under their own brand or a private label. The factory name rarely appears on the product page.
This matters for two reasons. First, pricing. A domestic distributor buying at container rates and reselling in pallet quantities is adding 25–40% to the landed factory cost before you see a price. If you are buying 50,000 boxes a year, that margin compression is real money. Second, specification control. When you buy through a distributor, the spec sheet you receive reflects what the distributor ordered — not necessarily what the factory is capable of. If you need a specific thickness tolerance, an accelerator-free formulation, or a particular AQL level, you need to be talking to the factory directly, or at minimum to a supplier who can give you factory-level documentation.
The post-Brexit compliance picture has added another layer of friction. Before 2021, CE marking under EU Medical Device Regulation was the standard for medical-grade nitrile gloves entering the UK. Post-Brexit, the UK Conformity Assessed (UKCA) mark became the UK-specific requirement — but the transition timeline has been extended multiple times, and as of 2024, CE-marked products continue to be accepted in Great Britain under a recognition arrangement. The practical implication: your supplier needs to hold CE marking at minimum, and ideally should be able to provide UKCA documentation or confirm their CE certification is issued by a UK-approved body. Many distributors cannot tell you which notified body issued their supplier's CE certificate, let alone whether it qualifies under the UK's post-Brexit recognition rules.
What EN 455 Compliance Actually Requires from Your Supplier
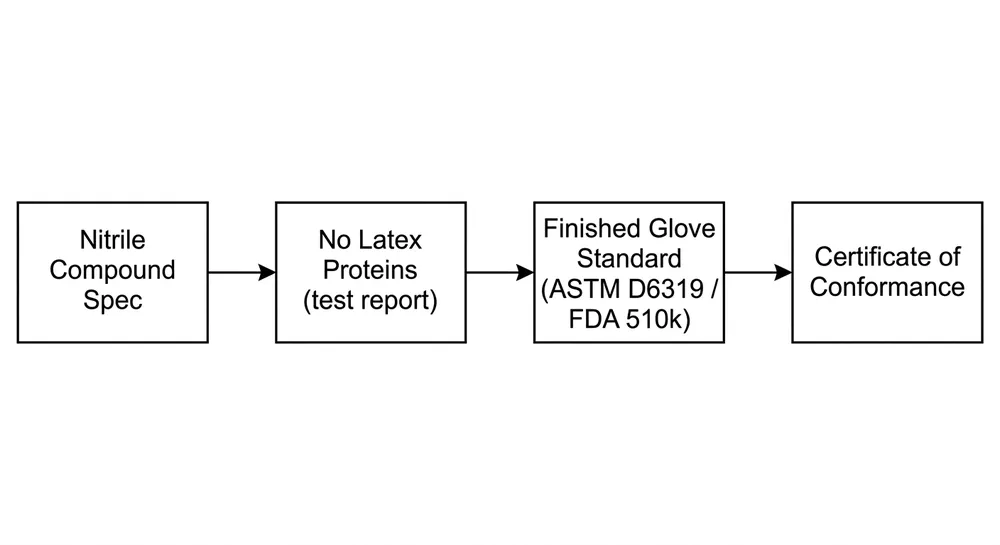
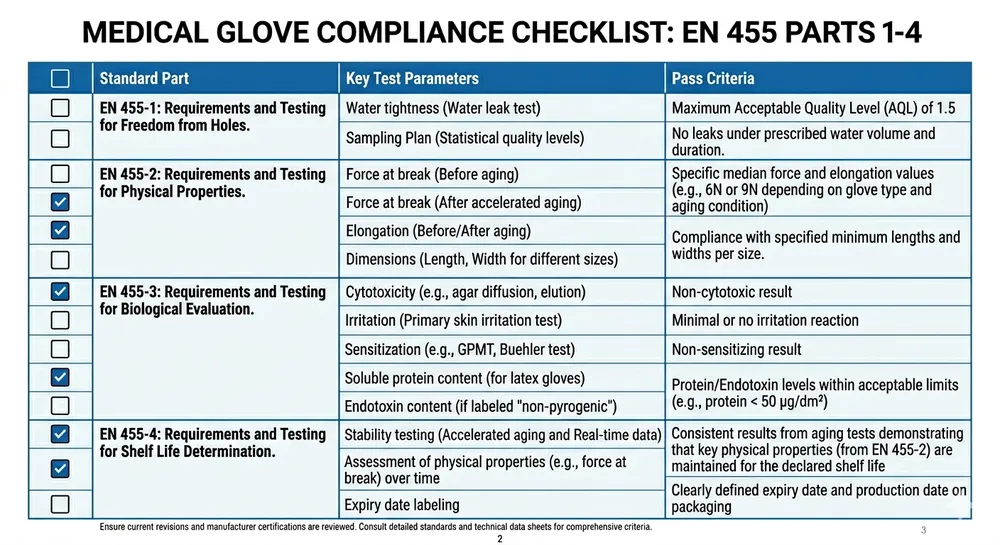
EN 455 is the European standard for single-use medical examination gloves. It has four parts, and all four matter for medical procurement:
- EN 455-1: Freedom from holes — AQL testing for pinholes using water-leak method
- EN 455-2: Physical properties — tensile strength, elongation at break, thickness
- EN 455-3: Biological evaluation — biocompatibility, residual powder levels, protein content
- EN 455-4: Shelf life determination — accelerated ageing and real-time ageing protocols
When a supplier says their gloves are "EN 455 compliant," the question to ask is: which parts, tested by which notified body, and when was the certificate last renewed? A certificate issued in 2019 against an older version of the standard may not satisfy current NHS procurement requirements. EN 455-2 was updated in 2015 and EN 455-3 in 2015 as well — certificates predating those revisions are not current.
For UK medical and dental procurement, the practical checklist is:
| Requirement | What to verify |
|---|---|
| EN 455-1 | AQL level for pinholes — AQL 1.5 is tighter than the standard's minimum |
| EN 455-2 | Tensile strength ≥6N before ageing, ≥4N after; elongation ≥500% |
| EN 455-3 | Powder-free confirmed; accelerator-free if NHS or dental use |
| EN 455-4 | Shelf life ≥3 years with supporting ageing data |
| CE marking | Notified body name and number on certificate |
| UKCA status | Confirm CE body is UK-approved or UKCA certificate available |
(We see a lot of certificates that pass EN 455-2 on tensile strength but have no post-ageing data attached. That gap matters if your buyer is storing product for 18 months before use.)
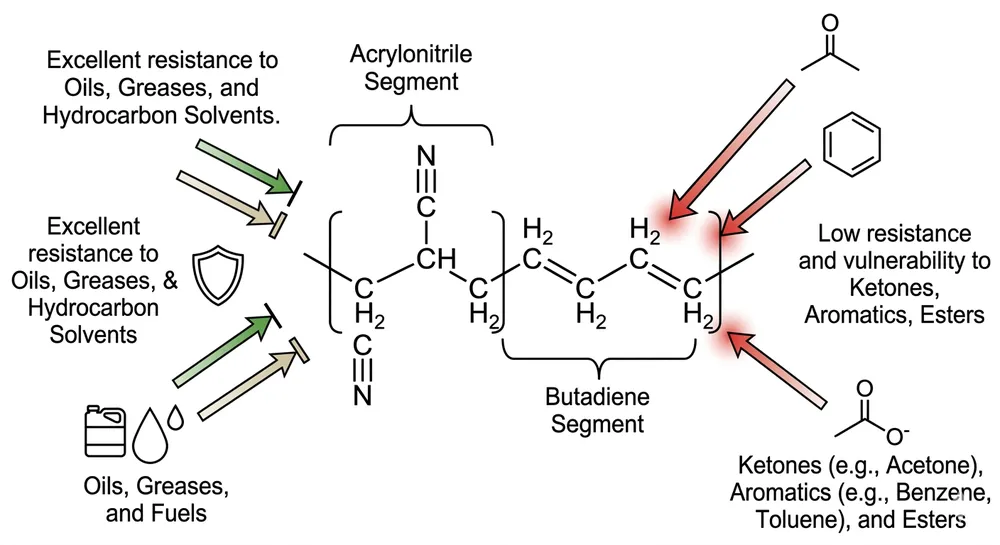
The accelerator-free requirement deserves specific attention for NHS and dental supply chains. Type IV latex allergy is well-documented, but accelerator chemicals used in nitrile vulcanisation — primarily thiurams and carbamates — are increasingly implicated in occupational contact dermatitis among healthcare workers. NHS procurement frameworks and dental practice groups are actively specifying accelerator-free gloves. A supplier without in-house compounding capability cannot reliably produce accelerator-free formulations — they are dependent on their raw material supplier's compound, which may change without notice.
Supplier Evaluation Criteria for UK Container Orders
Before reviewing specific suppliers, here is the evaluation framework that separates a reliable factory-direct source from a distributor with a factory tour photo on their website.
Certifications: CE (with notified body number), EN 455 parts 1–4, ISO 9001:2015. For US-parallel supply chains, FDA 510(k) and ASTM D6319 are additional signals of a factory operating to export-grade QC. A factory holding FDA 510(k) has been through a more rigorous documentation review than one with CE alone.
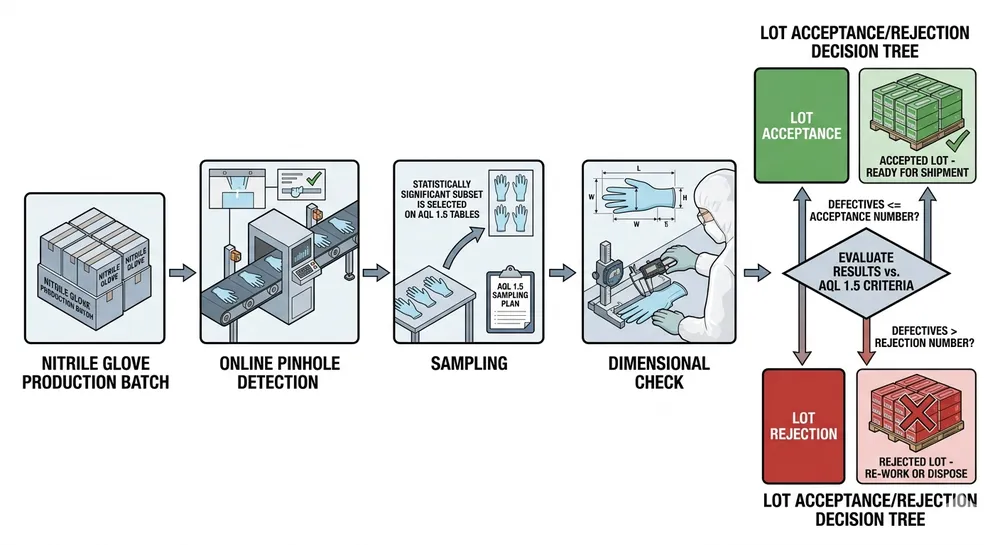
AQL level: The industry default for nitrile gloves is AQL 2.5 for pinholes. AQL 1.5 is a tighter standard — it means a smaller acceptable defect rate per batch. For medical and dental supply, AQL 1.5 outgoing inspection is the differentiator. Ask specifically: is AQL 1.5 applied at outgoing inspection, or only at incoming raw material? The answer tells you whether the factory is controlling quality at the point that protects your customer.
Capacity and line allocation: A factory running 4 lines at full capacity for domestic orders cannot reliably allocate line time to a new UK container account without displacing existing production. Ask for annual capacity figures and current utilisation. A factory with 1+ billion piece annual capacity and 8 lines has genuine scheduling flexibility. A factory with 200 million piece capacity and 4 lines is likely running at or near full utilisation.
Lead time with confirmed ship dates: The standard lead time for a container order from a Chinese factory is 25–35 days from order confirmation. What matters more than the headline number is whether the factory provides a confirmed ship date at order — not a range, not an estimate. Confirmed ship dates mean the factory has allocated line time before accepting your order.
MOQ and SKU flexibility: For UK importers building a product range, the ability to mix sizes within a container order (S/M/L/XL) without separate MOQ requirements per size is commercially important. A factory that requires 1 full container per size is effectively forcing you to overstock slow-moving sizes.
OEM and private label capability: If you are building a branded PPE range, the factory needs to handle custom box printing, logo placement, and colour-coded packaging. Ask for samples of existing OEM work, not just a yes/no answer.
10 Blue Nitrile Glove Suppliers Worth Evaluating for UK Importers
The suppliers below represent the realistic range of sourcing options for a UK importer. They are grouped by supplier model — domestic UK distributors, international trading companies, and factory-direct manufacturers — because the model determines what you can and cannot control in your supply chain.
UK-Based Distributors and PPE Wholesalers
1. Unigloves (UK) Website: unigloves.com Unigloves is one of the most established PPE brands in the UK market, with a dedicated blue nitrile range under their Pearl and Gold lines. They operate UK warehousing and offer next-day delivery on standard SKUs. Their gloves carry CE marking and EN 455 certification. For importers looking to white-label or buy at container scale directly, Unigloves operates as a brand rather than a factory — their manufacturing is outsourced to Asian production partners. Useful for urgent replenishment; less useful if you need factory-level specification control or OEM capability.
2. Polyco Healthline Website: polyco.com Polyco Healthline is a UK-headquartered PPE distributor with a broad nitrile glove range including their Finite and Bodyguards blue nitrile lines. They supply NHS trusts, food processing, and industrial accounts across the UK. Like Unigloves, they are a brand and distribution operation rather than a manufacturer. Strong on local stock depth and account management; limited on factory-direct pricing or custom specification work.
3. Supermax Healthcare (UK distribution) Website: supermaxhealthcare.com Supermax is a Malaysian manufacturer with a UK distribution presence. Their gloves carry CE, EN 455, and FDA certifications. As a manufacturer-owned distribution operation, they offer more factory-level documentation than a pure UK distributor — you can obtain batch-level test reports and traceability back to the production line. Lead times for non-stock items run longer than domestic distributors, but the certification depth is stronger.
4. Ansell (UK) Website: ansell.com Ansell is a global PPE manufacturer with a significant UK market presence. Their TouchNTuff and Micro-Touch nitrile lines are widely specified in healthcare and laboratory settings. Ansell operates at the premium end of the market — their pricing reflects brand positioning and global distribution infrastructure. For buyers who need a globally recognised brand name on the box, Ansell is a credible option. For buyers optimising landed cost on commodity-grade blue nitrile, the premium is hard to justify.
5. Shield Scientific Website: shieldscientific.com Shield Scientific supplies laboratory and cleanroom-grade nitrile gloves to UK research institutions, pharmaceutical manufacturers, and NHS laboratories. Their blue nitrile range is positioned at the technical end of the market — thicker gauges, tighter AQL, and cleanroom packaging options. Less relevant for standard medical examination or food-contact procurement; highly relevant if your buyer base includes pharmaceutical or life sciences accounts.

International Trading Companies and Sourcing Agents
6. Globus Group Website: globusgroup.co.uk Globus Group is a UK-based PPE company that sources and distributes nitrile gloves under their Bodyguards brand, with manufacturing partnerships in Asia. They have supplied NHS and local authority contracts and carry relevant UK compliance documentation. Their model sits between a pure distributor and a sourcing agent — they have more factory visibility than a typical UK wholesaler, but they are not a manufacturer. Useful for buyers who want UK-based account management with some factory-level documentation access.
7. Medline Industries (UK) Website: medline.com Medline is a US-headquartered medical supply company with UK operations. Their nitrile glove range covers medical examination, surgical, and industrial grades. For UK importers, Medline is primarily a supply channel rather than a factory-direct option — they source from multiple manufacturing partners and distribute under their own brand. Strong on compliance documentation for NHS procurement; less competitive on unit pricing for commodity-grade blue nitrile at container scale.
8. Rubberex (Malaysia) Website: rubberex.com Rubberex is a Malaysian nitrile glove manufacturer that sells directly to international importers including UK buyers. They hold CE, EN 455, and FDA certifications and offer OEM capability. As a manufacturer, they can provide factory-level documentation, batch test reports, and direct line allocation. Lead times from Malaysia to UK ports run 28–35 days. For UK importers comfortable with direct factory sourcing, Rubberex represents a credible Asian manufacturer alternative to Chinese supply.
9. Top Glove (international export) Website: topglove.com Top Glove is the world's largest glove manufacturer by volume, headquartered in Malaysia. They supply blue nitrile gloves to distributors and importers globally, including UK accounts. Their scale means pricing is competitive on commodity SKUs, and their certification portfolio is comprehensive. The trade-off is MOQ — Top Glove's direct export programme is oriented toward large-volume buyers, and smaller UK importers typically access their product through regional distributors rather than direct factory accounts.
10. Hartalega (Malaysia) Website: hartalega.com.my Hartalega is a Malaysian manufacturer known for thin-wall nitrile technology and high-volume automated production. Their NEXT Generation Integrated Glove Manufacturing Complex runs highly automated lines with consistent thickness tolerances. They supply to medical and industrial markets globally. Like Top Glove, direct access for smaller UK importers is limited — their primary channel is through appointed distributors. For buyers with sufficient volume to qualify for a direct account, Hartalega's technical capabilities are strong.
The Cost of Convenience: What UK Distributor Pricing Actually Includes
Buying from a UK distributor is the right answer in specific situations: urgent orders under 50,000 boxes, emergency replenishment when a container is delayed, or when your customer needs next-day delivery and you cannot hold stock. Local convenience has genuine value, and it is worth paying for when the situation demands it.
The question is whether you are paying for convenience you actually need, or paying for it by default because the factory-direct alternative feels complicated.
A UK distributor buying blue nitrile gloves at container rates from a Chinese or Malaysian factory is typically paying £2.80–£3.80 per box (100 gloves, powder-free, standard medical grade) depending on thickness and certification level. By the time that product reaches you at pallet quantities, the price is typically £4.50–£6.50 per box, depending on the distributor's margin structure and your account volume. That spread — £1.70 to £2.70 per box — is the cost of the distribution layer.
At 100,000 boxes per year, that spread is £170,000–£270,000 in additional cost versus buying at or near factory-direct landed cost. At 50,000 boxes, it is £85,000–£135,000. These are not precise figures — they are representative of the margin structure in this supply chain, and your actual numbers will depend on specification, volume, and supplier. But the order of magnitude is real, and it is the reason serious importers at repeat volume eventually move toward factory-direct sourcing.
(The other cost that rarely appears in a distributor's price is specification drift. When a distributor switches their factory source — which happens more often than buyers realise — the product in the box may change without the SKU changing. Factory-direct sourcing eliminates that risk because you are specifying directly with the manufacturer.)
The landed cost calculation for a factory-direct container order from China to a UK port typically looks like this:
| Cost component | Approximate range |
|---|---|
| Factory price (FOB Qingdao) | £2.60–£3.60 per box |
| Ocean freight (20ft container, China–Felixstowe) | £1,800–£2,800 per container |
| UK import duty (nitrile gloves, HS 4015.19) | 2.7% on CIF value |
| UK VAT (medical-grade, zero-rated) | 0% for EN 455 medical examination gloves |
| Customs clearance and port handling | £350–£600 |
| Inland delivery to your warehouse | £150–£400 |
A 20ft container holds approximately 2,000 boxes of standard blue nitrile gloves (100-count, powder-free). At a factory price of £3.00 per box and total logistics cost of approximately £0.25–£0.35 per box, your landed cost per box is in the £3.25–£3.35 range — versus £4.50–£6.50 from a UK distributor at pallet quantities. The margin you recover funds your next container order.
Factory-Direct Sourcing: What to Verify Before You Commit
The risk in factory-direct sourcing is not the factory — it is the gap between the sample and the production run. We have seen this pattern enough times to know where it happens: the sample is pulled from a controlled batch, inspected carefully, and shipped to the buyer. The production run is pulled from a standard batch under normal line conditions. If the factory's QC process does not apply the same inspection standard to outgoing production as it does to samples, the container that arrives at your warehouse will not match what you approved.
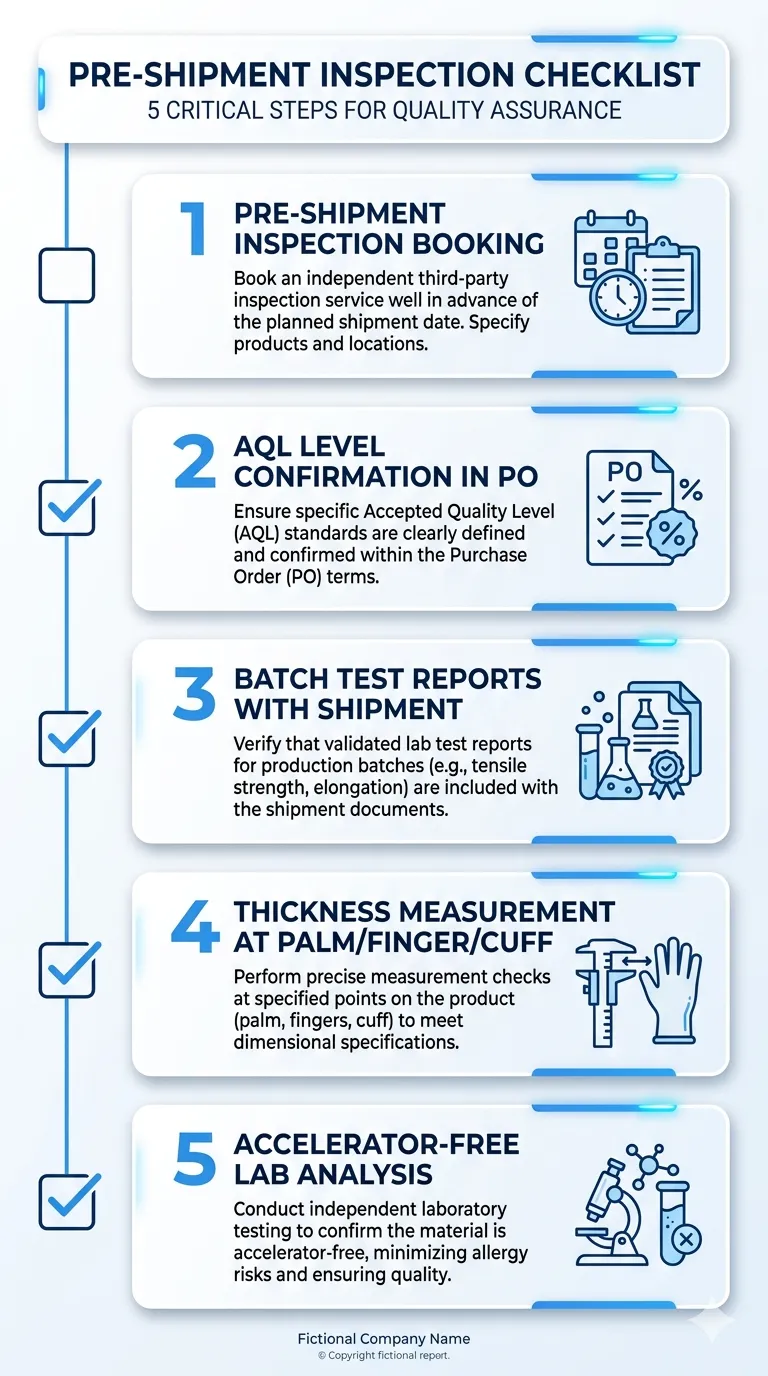
The verification steps that close this gap:
Pre-shipment inspection: Either your own inspector or a third-party inspection company (SGS, Bureau Veritas, Intertek) at the factory before the container is sealed. This is standard practice for serious importers and costs £300–£600 per inspection. It is cheap insurance against a rejected container.
AQL level confirmation in writing: Ask the factory to confirm in the purchase order that outgoing inspection is conducted at AQL 1.5 for pinholes and AQL 2.5 for physical properties. If they cannot confirm this in writing, they are not applying it consistently.
Batch test reports with shipment: Every container should be accompanied by batch-level test reports — not just a copy of the annual EN 455 certificate. Batch reports show the actual test results for the specific production run in your container.
Thickness measurement at multiple points: EN 455-2 specifies minimum thickness at the palm, finger, and cuff. Ask for the actual measured values from the production batch, not just "meets EN 455-2." A glove that measures 0.10mm at the palm and 0.07mm at the fingertip is technically within some interpretations of the standard but will fail at the thin point under stress.
Accelerator-free documentation: If you need accelerator-free gloves, ask for a chemical analysis report confirming the absence of thiurams, carbamates, and dithiocarbamates — not just a supplier declaration. The analysis should be from an accredited laboratory, not an internal factory test.
For more detail on how AQL standards work in practice for nitrile glove procurement, see our guide to AQL standards for nitrile gloves.
How glovemfg Serves UK Container Importers
We have been manufacturing nitrile gloves in Qingdao since 2007. The facility runs 8 fully automated dipping lines across 18,000 square meters, with 220 people on the floor and annual output at 1.2 billion pieces. We do not make latex gloves, vinyl gloves, or a mixed PPE catalog — only nitrile, across medical, industrial, and food-contact grades, from one facility.
For UK importers specifically, the certifications that matter are CE marking and EN 455 parts 1–4. We hold both, along with ISO 9001:2015, ASTM D6319, and FDA 510(k). The FDA 510(k) clearance is worth noting because it requires a more rigorous documentation review than CE alone — it means our QC system has been audited against a standard that most UK-focused factories do not bother with. For buyers who need to demonstrate supplier due diligence to NHS procurement or hospital group purchasing organisations, that documentation depth matters.
Our outgoing inspection runs at AQL 1.5 for pinholes — tighter than the EN 455-1 minimum and tighter than the AQL 2.5 standard most factories apply. We run 100% online pinhole detection on every line using automated water-inflation testing, and AQL 1.5 sampling at outgoing QC before container sealing. The practical result: our rejection rate at destination ports is below 0.3% across all export markets. (We track this because our UK and European buyers report back when they have issues — and the absence of reports is the data point we care about most.)
The accelerator-free capability is a genuine differentiator for NHS and dental procurement. We run an in-house compounding lab, which means we formulate our own nitrile compound rather than buying pre-mixed compound from a raw material supplier. That control lets us produce accelerator-free formulations — confirmed by third-party chemical analysis, not just a supplier declaration — without changing our production line setup. Most factories cannot do this because they do not own their compound formulation.
On lead time: we confirm ship dates at order, not at production start. A UK container order placed today gets a confirmed loading date within 48 hours of order confirmation. Standard lead time is 25–35 days from order to vessel departure from Qingdao. We do not quote ranges and then revise them — if we cannot meet a date, we say so before you place the order.
MOQ for standard blue nitrile SKUs is 1 container (approximately 2,000 boxes). Size mixing within a container is supported without separate per-size MOQ requirements — you can split a container across S/M/L/XL based on your sales mix. OEM and private label are available on the same container MOQ, including custom box printing and colour-coded packaging.
For UK importers building a blue nitrile gloves wholesale programme, the economics of factory-direct sourcing at container scale are straightforward. The certification depth, AQL standard, and lead time reliability are the variables that determine whether the first container becomes a repeat programme.
You can review our full Blue Nitrile Gloves product range, including thickness grades, accelerator-free options, and OEM specifications.
Which Sourcing Route Fits Your Situation
The honest answer is that the right sourcing model depends on your order pattern, not on which supplier has the best marketing.
Buy from a UK distributor when:
- You need delivery in under 5 days
- Your order is under 20,000 boxes and you cannot hold container-scale stock
- You are in an emergency replenishment situation
- You are testing a new SKU before committing to a container order
Move to factory-direct when:
- You are buying 50,000+ boxes per year on a predictable schedule
- You need EN 455 batch-level documentation for NHS or hospital procurement
- You need accelerator-free formulations that your current distributor cannot reliably source
- You are building a private-label PPE range and need OEM capability
- Your current distributor has had two or more specification inconsistency issues in the past 12 months
The hybrid model — buying from a UK distributor for urgent replenishment while running a factory-direct container programme for planned volume — is what most serious importers land on. The distributor relationship covers your emergency buffer; the factory-direct programme covers your margin.
The compliance question is worth addressing directly: post-Brexit UKCA/CE marking is not a reason to avoid factory-direct sourcing from China. A Chinese factory holding CE certification from a UK-approved notified body satisfies the current UK import requirements for medical examination gloves. The documentation you need is the CE certificate with the notified body number — your customs broker can confirm whether that body is on the UK's approved list. If you need UKCA marking specifically, ask the factory whether their CE certificate was issued by a UK-approved body, or whether they have initiated UKCA certification separately.
Frequently Asked Questions
What is the minimum order for a factory-direct container of blue nitrile gloves shipped to the UK? A standard 20ft container holds approximately 2,000 boxes of powder-free blue nitrile gloves (100-count). That is the practical MOQ for factory-direct sourcing from most Chinese manufacturers. Some factories require a full 40ft container (approximately 4,000 boxes) as their minimum — confirm this before requesting a price. At glovemfg, the MOQ is 1 × 20ft container with size mixing supported within that container.
Do EN 455-certified gloves from China meet UK import requirements post-Brexit? Yes, provided the CE certificate was issued by a notified body that is either UK-approved or recognised under the current UK-EU mutual recognition arrangement. The UK has extended acceptance of CE-marked medical devices for Great Britain through a transitional period. Your customs broker or a UK regulatory consultant can confirm the current status for your specific product category. The key document to request from your supplier is the CE certificate with the notified body name and number — not just a declaration of conformity.
What is the difference between AQL 1.5 and AQL 2.5 for nitrile gloves, and does it matter for UK procurement? AQL 2.5 means that in a standard sampling inspection, a batch with up to 2.5% defective units may be accepted. AQL 1.5 applies a tighter acceptance threshold — approximately 40% fewer defects accepted per batch. For NHS procurement and hospital group purchasing, AQL 1.5 is increasingly specified in tender documents. For industrial and food-contact procurement, AQL 2.5 is typically sufficient. The practical difference shows up at destination port inspection — a batch that passes AQL 2.5 at the factory may fail a buyer's incoming inspection at AQL 1.5. See our detailed guide to AQL standards for nitrile gloves for the full breakdown.
Are accelerator-free blue nitrile gloves available from factory-direct suppliers? Yes, but not from all factories. Accelerator-free formulations require the factory to control their own nitrile compound — factories that buy pre-mixed compound from a raw material supplier cannot reliably produce accelerator-free gloves because the compound formulation is outside their control. Ask specifically whether the factory runs an in-house compounding lab and whether they can provide third-party chemical analysis confirming the absence of thiurams and carbamates — not just a supplier declaration.
What documentation should I request for a UK container import of medical-grade blue nitrile gloves? At minimum: CE certificate with notified body number, EN 455 parts 1–4 test reports (batch-level, not just annual certificate), ISO 9001:2015 certificate, commercial invoice, packing list, bill of lading, and a certificate of origin. For NHS or hospital procurement, also request: AQL inspection report for the specific production batch, accelerator-free chemical analysis if applicable, and shelf life documentation per EN 455-4. Your freight forwarder will handle the customs entry, but the compliance documentation is your responsibility to collect from the supplier before the container ships.
To discuss container pricing, EN 455 documentation, or accelerator-free specifications for your UK import programme, Request Quote with your grade, size range, and annual volume — we will return a container-level price and confirmed lead time within 48 hours.