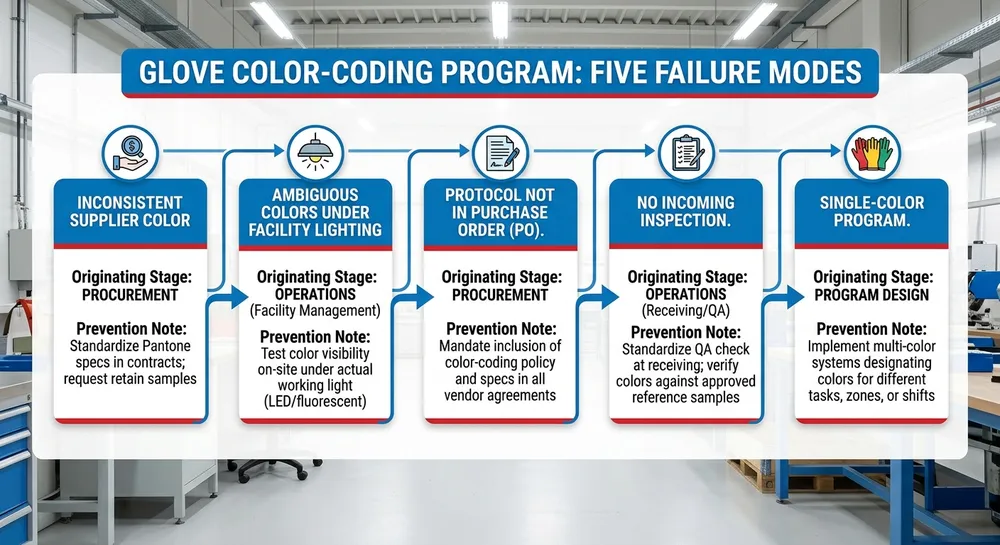
Latex-Free Blue Nitrile Gloves and Allergy Compliance: What US Healthcare Procurement Needs to Document
The compliance failure we see most often isn't a bad glove. It's a procurement file that can't prove the glove is what the label says it is.
A hospital system gets audited. The infection control officer asks for documentation confirming the exam gloves are latex-free. The distributor calls the importer. The importer calls the trading company. The trading company calls the factory — and somewhere in that chain, the original test report either doesn't exist, covers a different lot, or uses a standard the auditor doesn't recognize. The gloves are fine. The paperwork isn't. That's a compliance failure, and it's entirely avoidable if you know what to collect before the container ships.
This article covers what "latex-free" actually means at the manufacturing level, which documents US healthcare procurement needs to hold, and where the latex-free vs. accelerator-free distinction matters — because conflating those two is the most common documentation gap we see in hospital and clinic procurement files.
—
What "latex-free" means at the formulation level — and why the label alone isn't enough
"Latex-free" on a glove box is a marketing claim. What makes it a verifiable compliance claim is the formulation and the test data behind it.
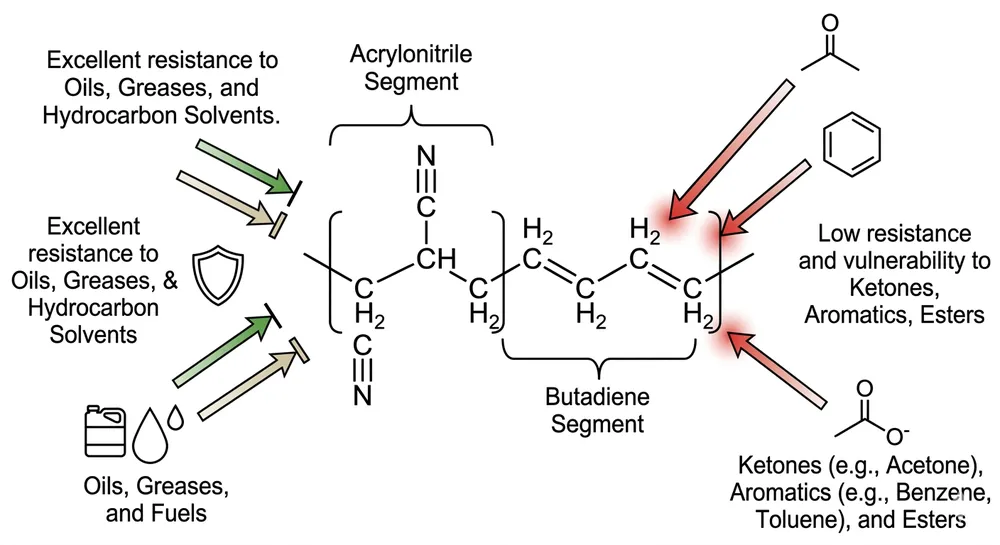
Nitrile gloves are made from acrylonitrile-butadiene rubber — a fully synthetic polymer with no natural rubber latex in the compound. The latex allergy risk (Type I hypersensitivity, IgE-mediated) comes from proteins in Hevea brasiliensis natural rubber. A properly formulated nitrile glove contains none of those proteins because the base polymer is synthetic from the start.
The operative phrase is "properly formulated." In our compounding lab, we mix the nitrile compound in-house, which means we control exactly what goes into the bath. There's no natural rubber latex in our nitrile compound, no latex-containing processing aids, and no shared equipment with latex production — we've never run latex on these lines. That's a formulation-level guarantee, not a label claim.
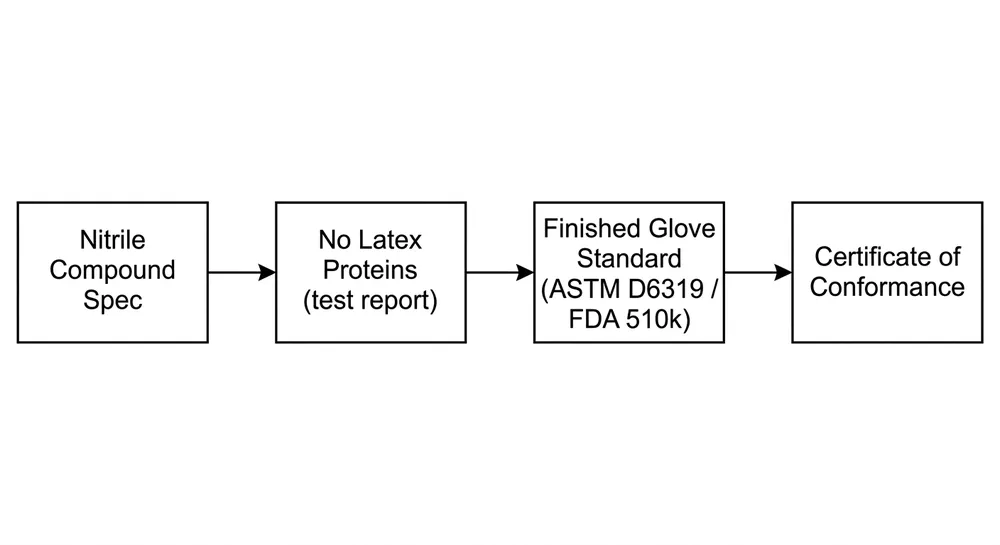
What procurement needs to document is the chain of evidence: the compound specification, the test report confirming absence of latex proteins, and the certification that the finished glove meets the applicable standard. A label that says "latex-free" without that chain is not sufficient for a hospital procurement audit.
—
The compliance documentation stack US healthcare procurement needs to hold
When a hospital group, dental network, or clinic procurement office sources exam gloves, the documentation requirement isn't just "are these certified?" It's "can you produce the specific documents that prove this specific lot is compliant?" Those are different questions.
Here's what the file should contain:
FDA 510(k) clearance reference For exam gloves sold into US healthcare, FDA 510(k) clearance is the baseline. It confirms the glove has been reviewed as a Class II medical device and cleared for the intended use. The 510(k) number should appear on the product label and in the supplier's documentation package. Our Latex Free Blue Nitrile Gloves carry FDA 510(k) clearance — the number is available in the documentation package we provide with every order.
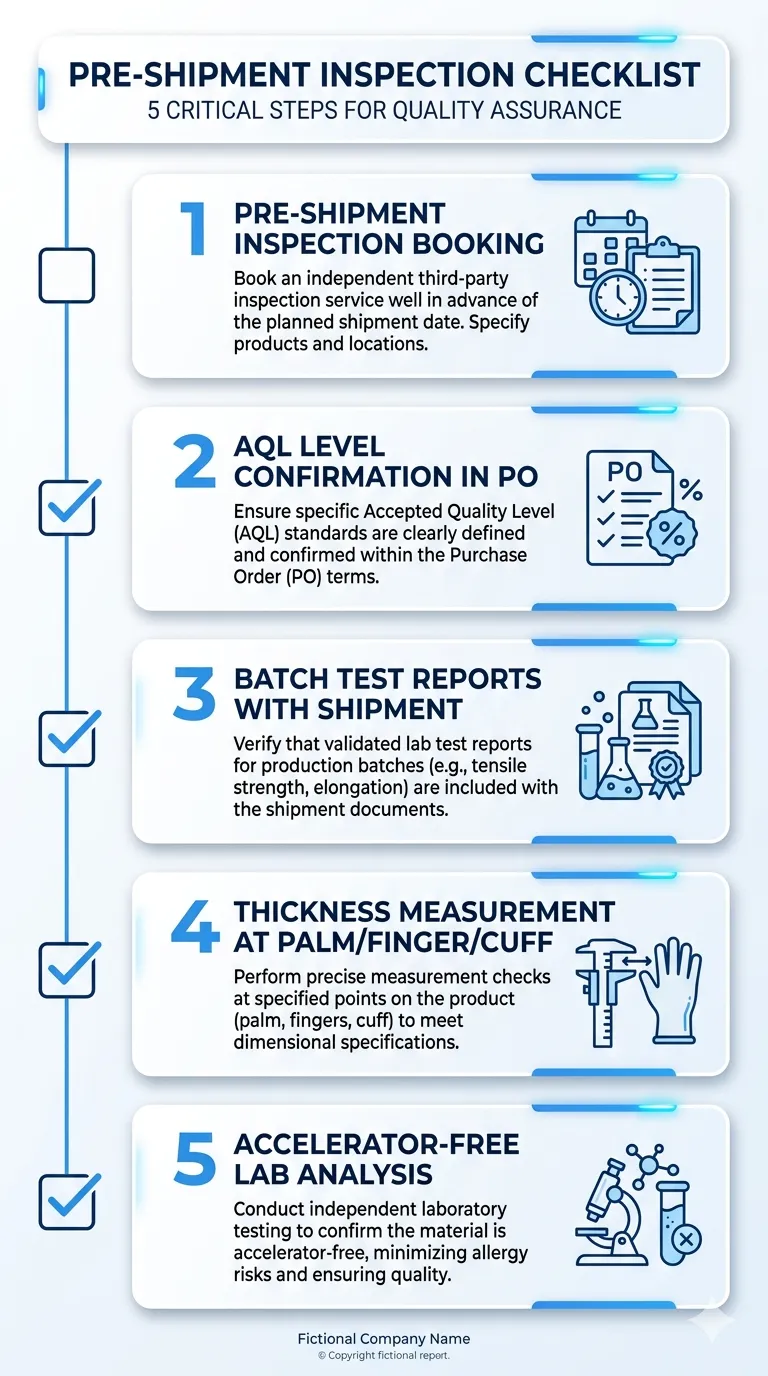
ASTM D6319 test report ASTM D6319 is the US standard for nitrile rubber examination gloves. It covers physical dimensions, tensile strength, elongation at break, and freedom from holes (AQL). A current test report to ASTM D6319 from an accredited third-party lab is what an FDA import reviewer or hospital compliance officer will ask for. The report should reference the specific product SKU and lot range it covers — a generic report from three years ago on a different SKU doesn't satisfy this requirement.
Certificate of Conformance (CoC) The CoC is the supplier's written declaration that the specific shipment lot meets the stated specifications and applicable standards. It should reference the lot number, production date, applicable standards (ASTM D6319, FDA 510(k) number), and the specific product description. A CoC that says "nitrile gloves, latex-free" without lot traceability is not useful for an audit.
Lot traceability record This is the link between the CoC and the physical product in your warehouse. Each carton should carry a lot number that traces back to a specific production batch, with corresponding QC records. If a compliance question arises six months after delivery, you need to be able to pull the production record for that lot — not just the general certification for the product line.
| Document | What it proves | Who asks for it |
|---|---|---|
| FDA 510(k) clearance | Device cleared for US medical use | Hospital procurement, FDA import review |
| ASTM D6319 test report | Physical performance to US exam glove standard | Compliance audits, distributor qualification |
| Certificate of Conformance | This specific lot meets stated specs | Hospital procurement, GPO audits |
| Lot traceability record | Physical product links to QC records | Recall readiness, compliance investigations |
| Latex protein test report | No detectable natural rubber latex proteins | Allergy compliance documentation |
We provide all five with every order. That's not a special request — it's the standard documentation package because our buyers' customers ask for it.
—
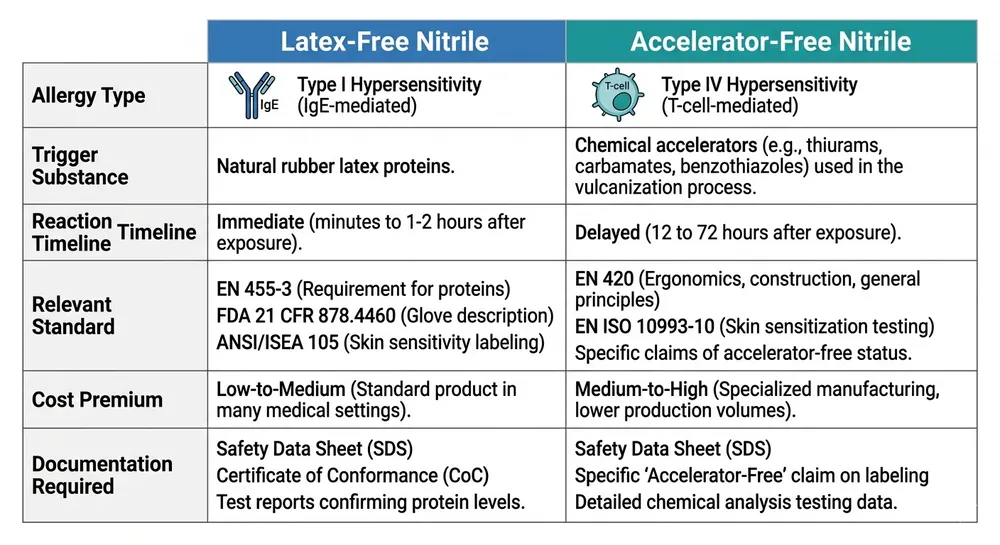
Latex-free vs. accelerator-free: two distinct allergy risks that procurement must address separately
This is the distinction that causes the most confusion in procurement files, and conflating them creates a real compliance gap.
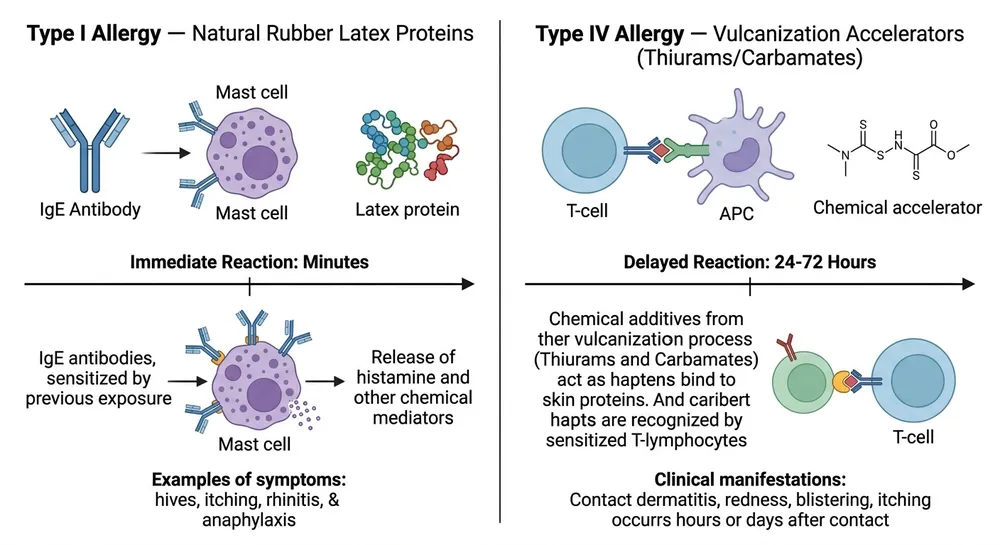
Type I latex allergy is an IgE-mediated immune response to natural rubber latex proteins. It can cause urticaria, rhinitis, asthma, and in severe cases anaphylaxis. The trigger is the latex protein itself. A nitrile glove with no natural rubber latex in the formulation eliminates this risk entirely — that's what "latex-free" addresses.
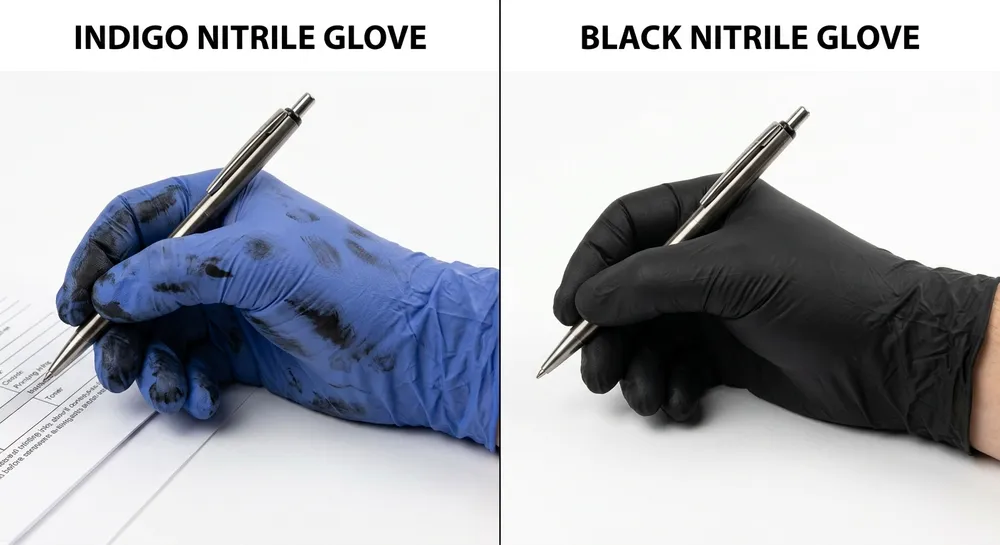
Type IV contact dermatitis is a delayed hypersensitivity reaction, not to latex proteins, but to the chemical accelerators used in the vulcanization process. The primary culprits are thiuram compounds and carbamate compounds — both are standard accelerators in conventional nitrile glove manufacturing. A glove can be completely latex-free and still trigger Type IV reactions in sensitized individuals.
The practical implication: a healthcare worker who reports a "latex glove reaction" may actually be reacting to accelerator residues in a nitrile glove, not to latex proteins. If your procurement file documents latex-free status but doesn't address accelerator content, you haven't fully covered the allergy compliance question for sensitized staff.
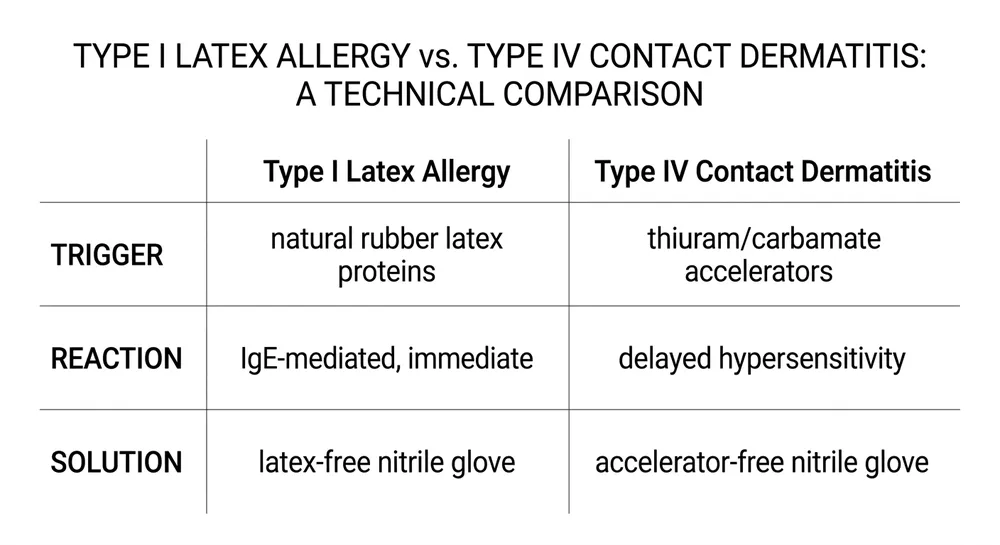
For procurement serving facilities with documented Type IV cases, or where staff have reported reactions to nitrile gloves, the specification needs to go one step further: accelerator-free nitrile.
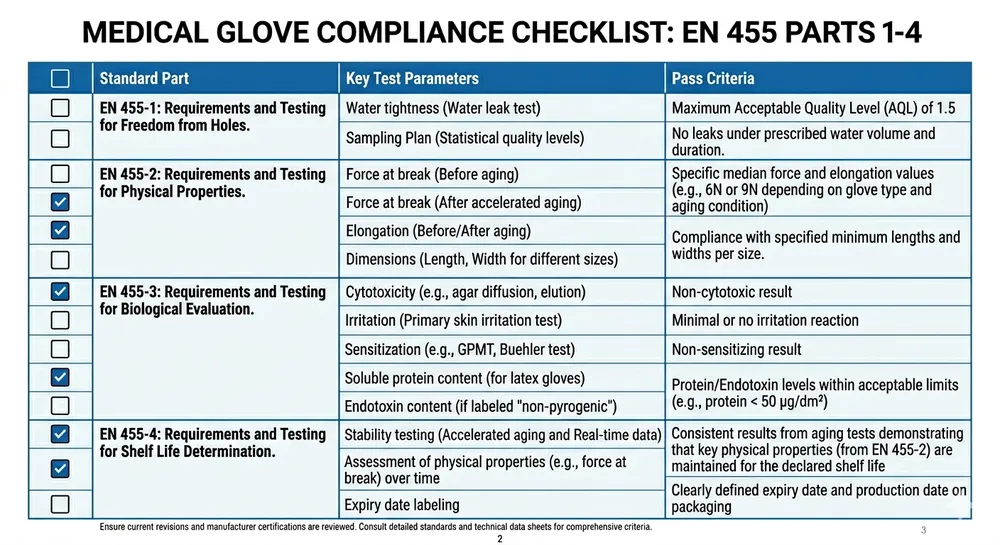
We run accelerator-free compound on a dedicated line using a sulfur-donor cure system instead of thiuram/carbamate accelerators. The finished glove carries no detectable accelerator residue and passes EN 455 testing. For US buyers, we can provide the corresponding test documentation alongside the standard ASTM D6319 and FDA 510(k) package. (This variant has been in our standard production rotation since 2019 — it's not a custom project, it's a stocked SKU.)
For a deeper look at how these two specifications differ and when each applies, see our article on latex-free vs accelerator-free nitrile gloves.
—
How to verify latex-free status across repeat container orders, not just the initial sample
The sample shipment is the easy part. The compliance question that actually matters is whether the 10th container, ordered 14 months after the first, is the same glove with the same formulation and the same documentation.
This is where factory-direct sourcing and in-house compounding make a material difference.
When you source through a trading company or multi-tier distributor, the documentation chain has gaps at every handoff. The trading company may have the original factory's test report, but they may not know whether the factory changed compound suppliers, adjusted the formulation, or subcontracted production to a second facility. We've seen procurement files where the CoC on file was from the original qualification sample, and the subsequent containers came from a different production source entirely — same label, different glove.
At our facility, the nitrile compound is mixed in-house from the same formulation specification that was used for the original qualification. We don't buy pre-mixed compound from a third-party supplier, so there's no upstream formulation change that happens without our knowledge. When you reorder, the compound specification is the same, the production line is the same, and the documentation package covers the new lot number — not a reissue of the original sample report.
What to require from any supplier for repeat orders:
- Lot-specific CoC for each shipment — not a blanket certificate covering the product line
- Current ASTM D6319 test report — ideally within the past 12–18 months, referencing the current SKU
- Compound specification confirmation — written confirmation that the formulation has not changed since the original qualification
- Production facility declaration — confirmation that the goods were produced at the same facility as the qualified sample, not subcontracted
(We include lot-specific CoC and production facility declaration as standard in every shipment documentation package. If a buyer needs compound specification confirmation in writing, we provide it on request — it's a one-page document from the compounding lab.)
—
What to require from a manufacturer to satisfy hospital and clinic procurement audits
Hospital group purchasing organizations (GPOs) and individual facility procurement offices have become more rigorous about supplier qualification since 2020. The documentation requirements have moved beyond "show me the certification" toward "show me the quality system behind the certification."
Here's what a well-prepared supplier qualification file looks like for latex-free exam gloves in US healthcare procurement:
Regulatory clearance
- FDA 510(k) clearance number, product name, and cleared indications
- Current FDA establishment registration number for the manufacturing facility
Product performance
- ASTM D6319 test report from an accredited third-party lab (current, lot-referenced)
- AQL inspection standard used (AQL 1.5 is the tighter standard; AQL 2.5 is more common but less rigorous)
- Pinhole detection method — electrical leak test on 100% of production is the standard we use; visual-only inspection is not equivalent
Allergy compliance
- Latex protein test report confirming no detectable natural rubber latex
- Accelerator content declaration (or accelerator-free test report if that specification is required)
- Powder-free confirmation — powder-free is now the standard for medical exam gloves; powdered latex gloves are FDA-banned for medical use, and powdered nitrile, while not banned, is not appropriate for clinical settings
Quality system
- ISO 9001:2015 certificate with current validity date
- Most recent audit summary or surveillance audit confirmation
Supply chain
- Manufacturing facility address and confirmation that production is not subcontracted
- Lot traceability procedure — how lot numbers link to production records
Our Blue Nitrile Gloves documentation package covers all of these. ISO 9001:2015, CE, EN 455, ASTM D6319, and FDA 510(k) are current certifications. AQL 1.5 is our outgoing inspection standard across all 8 lines. Online electrical pinhole detection runs on every line. Production is not subcontracted — everything ships from our Qingdao facility.

—
The sourcing trap: documentation gaps that cause compliance failures at audit time
Most compliance failures in healthcare glove procurement aren't discovered at the port. They surface six months later when a facility gets audited, a staff member reports a reaction, or a GPO runs a supplier qualification review.
The most common gaps we see:
Generic certifications without lot traceability. A supplier provides an FDA 510(k) number and an ASTM D6319 test report, but neither document references the specific lot in the warehouse. If the auditor asks "prove this box came from a compliant production run," the answer is in the lot number — and if the lot number doesn't appear in the documentation, the file fails.
Outdated test reports. ASTM D6319 test reports don't expire on a fixed schedule, but a report from five years ago on a product that has since been reformulated or moved to a different production facility is not evidence of current compliance. Procurement should confirm the test report date and ask whether the formulation or production source has changed since the report was issued.
Latex-free label without protein test data. "Latex-free" on the box is a manufacturer's claim. The supporting evidence is a latex protein test report — typically using ELISA or similar immunoassay method — confirming no detectable natural rubber latex proteins in the finished glove. Without that report, the claim is unverifiable.
Accelerator content not addressed. As covered above, a latex-free glove can still contain thiuram and carbamate accelerators. If your facility has staff with documented Type IV sensitivity, or if your GPO contract requires accelerator-free specification, a latex-free CoC alone doesn't cover the requirement.
Trading company CoC instead of manufacturer CoC. A CoC issued by a distributor or trading company is a pass-through document — it reflects what the trading company was told, not what the factory produced. For hospital procurement audits, the CoC should come from the manufacturing facility, not from an intermediary.
Factory-direct sourcing eliminates the last gap entirely. When you source from us directly, the CoC comes from the facility that made the gloves, the lot number traces to our production records, and there's no intermediary who may or may not have the original documentation. If an auditor asks a question about a specific lot, we can pull the production record, the QC inspection data, and the compound batch record for that run.
—
Frequently asked questions
Does "powder-free" mean the same thing as "latex-free"?
No. These are independent specifications. Powder-free means the glove was manufactured without cornstarch or other powder lubricants — relevant for sterile field use and for reducing airborne latex protein dispersal in latex gloves. Latex-free means the glove contains no natural rubber latex. A nitrile glove is both powder-free and latex-free by default if it's properly formulated, but the two claims address different things and require separate documentation.
Can a nitrile glove cause a latex allergy reaction?
A properly formulated nitrile glove with no natural rubber latex in the compound cannot trigger a Type I latex allergy reaction — there are no latex proteins present. However, nitrile gloves can trigger Type IV contact dermatitis reactions in individuals sensitized to thiuram or carbamate accelerators. If a healthcare worker reports a reaction to nitrile gloves, the likely cause is accelerator sensitivity, not latex allergy. The solution is accelerator-free nitrile, not a different glove material.
What is the difference between ASTM D6319 and EN 455 for procurement purposes?
ASTM D6319 is the US standard for nitrile rubber examination gloves, covering physical dimensions, tensile properties, and AQL for holes. EN 455 is the European standard for medical gloves for single use, covering similar performance parameters plus biological evaluation requirements. For US healthcare procurement, ASTM D6319 and FDA 510(k) are the primary compliance anchors. EN 455 is relevant if you're also supplying European markets or if your GPO contract references it. Our gloves carry both, so the documentation covers either procurement requirement.
How often should ASTM D6319 test reports be renewed?
There's no regulatory-mandated renewal interval for ASTM D6319 test reports, but most hospital GPOs and procurement offices treat reports older than 2–3 years as requiring reconfirmation, particularly if there's been any change in formulation, production facility, or supplier. We issue updated test reports on a rolling basis and can provide a current report for any active SKU on request.
What's the minimum documentation package for a first container order?
For a first container of latex-free exam gloves into US healthcare distribution, the baseline package should include: FDA 510(k) clearance reference, current ASTM D6319 test report, lot-specific Certificate of Conformance, latex protein test report, and powder-free declaration. If accelerator-free specification is required, add the accelerator content test report. We provide all of this as standard — you don't need to request each document separately. Submit your specification requirements when you Request Quote and we'll confirm the documentation package at the same time.