If you're running a medical supply distribution business or stocking PPE for industrial accounts, you've already narrowed it down to two options. Latex has decades of clinical history. Blue nitrile has taken over most high-volume procurement in the last fifteen years. The question isn't which one sounds better — it's which one holds up across a full year of orders without creating downstream problems for your customers.
Here's the short answer: for high-volume medical and industrial distribution, blue nitrile is the lower-risk, more commercially durable choice. Latex still wins in specific surgical and fine-tactile applications, but for the bulk of examination, procedural, and industrial-grade volume, nitrile gives you fewer allergy complaints, more consistent supply, and a sourcing chain that doesn't depend on natural rubber commodity swings.
The rest of this article shows you exactly where that verdict holds and where it doesn't.
—
Head-to-Head: The Properties That Actually Drive Procurement Decisions
Before getting into scenarios, here's the comparison matrix that matters for buyers sourcing at container scale.
| Property | Blue Nitrile | Latex |
|---|---|---|
| Allergy risk | Near-zero (synthetic, latex-protein-free) | Type I hypersensitivity risk — up to 17% sensitization in high-exposure healthcare workers |
| Chemical resistance | Strong against oils, solvents, many acids | Moderate — degrades faster with hydrocarbon exposure |
| Puncture resistance | Higher — synthetic polymer structure | Moderate — natural rubber is more elastic but tears under sharp puncture |
| Tensile strength | Comparable to latex at equivalent thickness | Slightly higher elongation at break |
| Thickness range | 0.08–0.15mm (controllable in manufacturing) | Typically 0.10–0.18mm, less consistent across batches |
| Shelf life | 3–5 years (synthetic, stable) | 2–3 years (natural rubber degrades faster) |
| Supply chain stability | Stable — petrochemical feedstock | Volatile — natural rubber commodity pricing |
| Color coding | Blue is a standard visual identifier for nitrile | Natural off-white/tan; dyed latex less common |
| Regulatory compliance | FDA 510(k), CE, EN455, ASTM D6319 achievable | FDA and CE achievable, but latex-specific allergen labeling required in EU/US |

The allergy row is the one that ends most latex conversations at the procurement level. Once a hospital system or food processing facility has had an incident — or even a formal complaint — they move to nitrile and don't come back. We've seen this pattern consistently across our North American and European accounts over the past decade.
—
The Hidden Cost Latex Buyers Discover Too Late
The unit price comparison between latex and nitrile is closer than it used to be. But the real cost gap shows up downstream, and it's not on the invoice.
Allergen liability exposure. In the US and EU, latex gloves require specific allergen labeling. If your downstream customer — a clinic, a food plant, a lab — has a Type I latex allergy incident involving product you supplied, the liability chain runs back to the distributor. Nitrile eliminates that exposure entirely. For distributors building accounts in healthcare, that's not a minor compliance footnote — it's a material risk reduction.
Shelf life and inventory write-off risk. Natural rubber degrades. A container of latex gloves sitting in a warm warehouse for 18 months will show accelerated aging — brittleness, tackiness, reduced tensile strength. Nitrile holds its properties for 3–5 years under standard storage conditions, so your inventory turns don't have to race against material degradation. (We've had buyers come to us specifically after writing off a partial container of latex that sat too long in a Southeast Asian distribution hub — it's a real cost that doesn't show up in the per-box price.)
Supply price volatility. Natural rubber is a commodity. Latex glove pricing moves with rubber futures, weather events in Southeast Asian growing regions, and seasonal harvest cycles. Nitrile feedstock pricing is more stable and more predictable for annual contract negotiations. If you're quoting your customers fixed annual pricing, nitrile gives you a more defensible cost floor.
Regulatory friction in new markets. If you're expanding into EU markets or healthcare accounts that require EN455 compliance, latex gloves carry additional documentation requirements around residual protein content. Nitrile skips that layer entirely.
—
Where Latex Still Wins — and Why It Matters for Your SKU Mix
Latex isn't obsolete. There are specific applications where it genuinely outperforms nitrile, and if your accounts include these segments, you need to know where the line is.
Surgical and fine-tactile procedures. Latex has a higher elongation at break and a more natural tactile feel that some surgeons and dental professionals still prefer for fine motor work. Surgical-grade latex gloves remain in use in operating rooms where the clinical team has been screened for latex sensitivity. If you're supplying surgical suites rather than examination rooms, this matters.
High-stretch applications. Latex stretches further before tearing. In applications where the glove needs to conform tightly over irregular shapes — certain dental procedures, some laboratory handling tasks — latex's elasticity is a functional advantage.
Price-sensitive short-run procurement. In markets where latex is locally produced and nitrile is imported, latex can still win on landed cost for buyers who aren't concerned about allergen risk. This is more relevant in Southeast Asian domestic markets than in North American or European distribution.
For most high-volume medical examination, food processing, industrial maintenance, and laboratory supply accounts, none of these exceptions apply. The bulk of the market has moved to nitrile for good commercial reasons.
—
Application Showdown: Three Scenarios, Three Verdicts
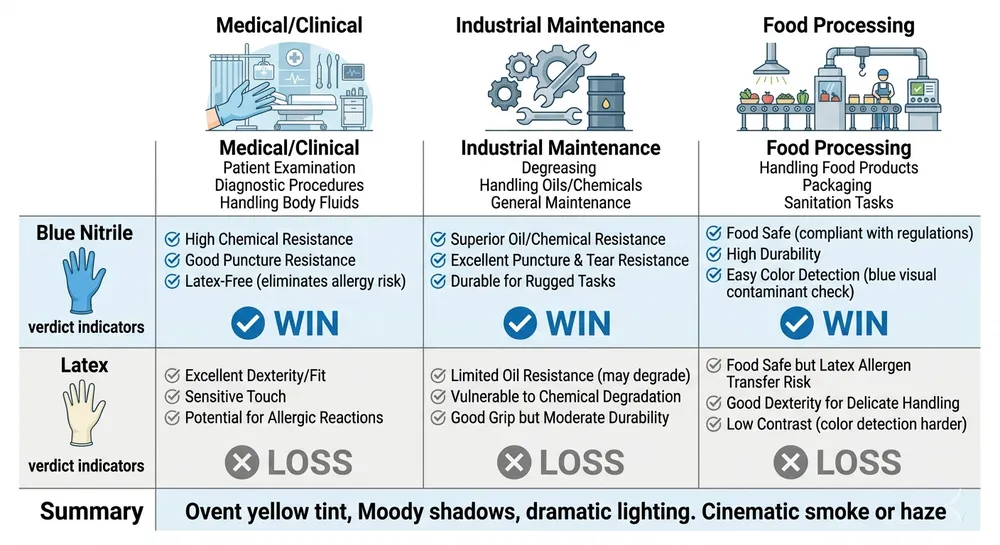
Scenario 1: Hospital System or Clinic Network — Examination Gloves at Scale
A regional hospital network buying 500,000–2,000,000 gloves per quarter for examination and procedural use.
Winner: Blue Nitrile — decisively.
Healthcare facilities have moved away from latex for examination use because the allergy risk is institutional, not individual. Staff rotate, patients have undisclosed sensitivities, and a single incident creates documentation, incident reporting, and potential liability. Blue nitrile eliminates that category of risk. The color coding also matters operationally — blue is a recognized visual identifier that helps facilities manage glove type segregation across departments. At this volume, the per-box price difference between nitrile and latex is negligible against the risk reduction value.
Scenario 2: Industrial Maintenance and Chemical Handling
A safety supply distributor stocking gloves for automotive workshops, manufacturing plants, and chemical handling environments.
Winner: Blue Nitrile — on chemical resistance and durability.
Latex degrades with hydrocarbon exposure — oils, greases, and many solvents attack natural rubber faster than they attack nitrile polymer. For mechanics, maintenance technicians, and chemical handlers, nitrile holds up longer per pair, which means your customer's per-use cost is lower even if the per-box price is similar. Puncture resistance is also higher in nitrile, which matters in environments with metal shavings, sharp edges, or abrasive surfaces. (Textured fingertip variants — which we produce on the same lines — add grip in oily conditions, which is a spec worth calling out to industrial accounts.)
Scenario 3: Food Processing and Packaging Lines
A food safety distributor supplying gloves to meat processing, bakery, and ready-meal production facilities.
Winner: Blue Nitrile — on color coding and contamination detection.
Blue is the standard color for food-contact gloves precisely because it's visually distinct from most food products. If a glove tears during processing, the blue fragment is detectable against most food backgrounds — a critical food safety control point. Latex in natural or off-white color fails this test. Beyond color, nitrile's chemical resistance handles the sanitizers and cleaning agents used in food processing environments better than latex. Most food safety auditors and HACCP programs now specify powder-free nitrile as the default — latex requires additional justification.
—
What to Verify Before the Container Ships
This is where most sourcing problems actually originate — not in the spec comparison, but in the gap between the spec sheet and what arrives at your warehouse.
AQL standard and inspection level. AQL 1.5 is the standard for medical-grade examination gloves. AQL 2.5 is common for industrial grade. Know which your account requires and confirm it in writing before the order. We run AQL 1.5 across all medical-grade lines with online pinhole detection — but not every factory does, and the difference shows up at destination port inspection, not before.
Thickness tolerance and consistency. A glove spec'd at 0.10mm palm thickness that measures 0.07mm at the fingertip fails AQL at the destination. Thickness variation is the most common spec drift issue between sample shipments and full container orders. Ask for thickness measurements at palm, finger, and cuff — not just a single-point average. Our 8 automated dipping lines hold 0.08–0.15mm tolerance across the full glove, which is what makes container-to-container consistency possible.
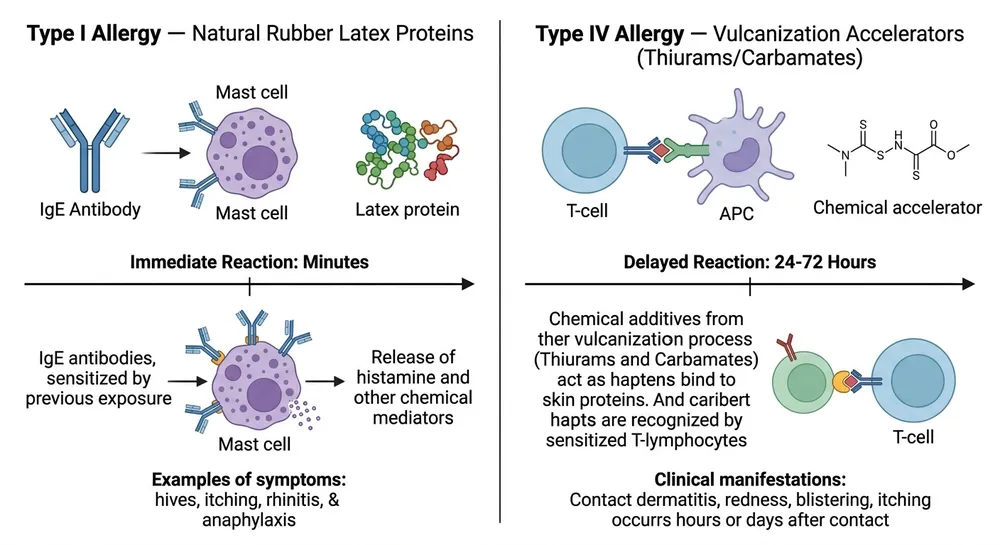
Accelerator-free certification if required. Standard nitrile gloves use chemical accelerators (thiurams, carbamates) in the vulcanization process. These can cause Type IV delayed hypersensitivity reactions — different from latex allergy but still a clinical concern for high-exposure healthcare workers. If your medical accounts are specifying accelerator-free gloves, confirm this explicitly. We produce accelerator-free variants from the same facility — it's a formulation change, not a different product line.
Powder-free verification. All medical-grade nitrile should be powder-free. Confirm this is documented in the product certificate, not just stated verbally. Powdered gloves are banned for medical use in the US (FDA 2017 ban) and restricted in EU medical device regulations.
Color consistency across batches. Blue nitrile color is not standardized across manufacturers — the shade varies by formulation. If your accounts use blue for color-coded contamination control (food processing, pharmacy), confirm the color standard and request batch-to-batch color consistency documentation. This is a detail that gets missed in initial RFQs and causes complaints six months into a supply relationship.
Certifications relevant to your market. For US distribution: FDA 510(k) for medical examination gloves, ASTM D6319 for industrial nitrile. For EU: CE marking under EN455 for medical, EN374 for chemical protection. Confirm the certificate number is current and the scope covers the specific SKU you're ordering — not just the factory's general certification.
—
Sourcing Blue Nitrile at Container Scale: What the Spec Sheet Doesn't Tell You
The comparison between nitrile and latex is straightforward once you understand the commercial logic. The harder part is finding a nitrile supplier who can hold spec across a full year of orders — not just the first sample shipment.
We've been manufacturing nitrile gloves exclusively since 2007. The facility runs 8 fully automated dipping lines across 18,000 square meters, with an annual output of 1.2 billion pieces. When you're placing quarterly container orders, that capacity means your order isn't competing with someone else's for line time.
The in-house compounding lab is what makes thickness consistency possible at scale. We control the nitrile formulation — we're not buying pre-mixed compound and hoping the batch is consistent. When a buyer reports thickness variation between their first and third container, the root cause is almost always compound variability. Owning the formulation is how we prevent that.
Standard SKUs ship from 1 container (approximately 2,000 boxes). OEM and private-label orders — custom color, custom branding, accelerator-free formulation — are supported from the same lines. ISO 9001:2015, CE, EN455, ASTM D6319, and FDA 510(k) certifications cover the full medical and industrial grade range.
If you're currently sourcing latex and evaluating a switch to nitrile, or if you're adding a blue nitrile SKU to an existing PPE line, the Blue Nitrile Gloves category page covers the full specification range — thickness options, grade classifications, and available certifications.
—
Frequently Asked Questions
Are blue nitrile gloves truly latex-free? Yes. Blue nitrile gloves are made from synthetic nitrile butadiene rubber — no natural latex proteins. They are safe for users with Type I latex hypersensitivity. This is the primary clinical reason most healthcare facilities have switched from latex examination gloves to nitrile over the past 15 years.
What thickness of nitrile glove is equivalent to standard latex examination gloves? Standard latex examination gloves typically run 0.10–0.12mm at the palm. Nitrile at the same thickness range (0.10–0.12mm) provides comparable barrier protection with higher puncture resistance. For industrial use where durability is the priority, 0.13–0.15mm nitrile is the more common spec.
Can nitrile gloves replace latex for surgical use? Surgical-grade nitrile gloves exist and are used in latex-sensitive environments, but standard examination-grade nitrile is not a direct surgical substitute. Surgical gloves require sterile packaging, higher elongation specifications, and specific tactile sensitivity standards. If your accounts include surgical supply, confirm the surgical-grade certification separately.
What does AQL 1.5 mean in practice for a distributor? AQL 1.5 means that in a standard sampling inspection, the acceptable quality level allows no more than 1.5 defects per 100 units. For a 10,000-glove lot, that translates to a maximum of 150 defective units before the lot fails inspection. AQL 2.5 — common for industrial grade — allows 2.5 defects per 100. The difference matters if your accounts are medical facilities with strict incoming inspection protocols.
How do I verify a factory's FDA 510(k) clearance covers the specific glove I'm ordering? The FDA 510(k) database is publicly searchable at fda.gov. Look up the clearance number the factory provides and confirm the device description matches the product type (examination glove, nitrile, powder-free) and that the clearance is not expired or withdrawn. A factory can hold 510(k) clearance for one SKU and sell you a different one — always match the clearance number to the specific product in your purchase order.