Every nitrile glove sold today is latex-free. That's not a premium feature — it's the baseline. Nitrile is a synthetic rubber; it contains no natural rubber latex proteins by definition. So when a supplier lists "latex-free" on a spec sheet, they're telling you something that's already true of every nitrile glove in the market.
The problem is that "latex-free" has become a proxy for "allergy-safe" in procurement language — and that conflation is costing healthcare facilities, dental practices, and food-processing operations real money in worker compensation claims, compliance failures, and emergency glove replacements mid-contract.
The allergy risk that actually affects nitrile glove users isn't from latex. It's from the chemical accelerators used to vulcanize the nitrile compound during manufacturing. These are two entirely different biological mechanisms, two different affected populations, and two different specifications. If you're sourcing gloves for an environment where Type IV sensitivity is a documented risk — and in European hospital tenders, it increasingly is a documented requirement — specifying "latex-free nitrile" without asking about accelerator content is specifying the wrong thing.
Here's the short version before we go deeper: latex-free nitrile addresses Type I (IgE-mediated) allergy to natural rubber proteins. Accelerator-free nitrile addresses Type IV (T-cell mediated) delayed hypersensitivity to vulcanization chemicals. A glove can be both, either, or — in the case of standard nitrile — neither concern addressed beyond the baseline.
—
Type I vs Type IV: The Allergy Mechanism Your Spec Sheet Needs to Reflect
The distinction matters because the two reactions have different triggers, different timelines, and different regulatory responses.
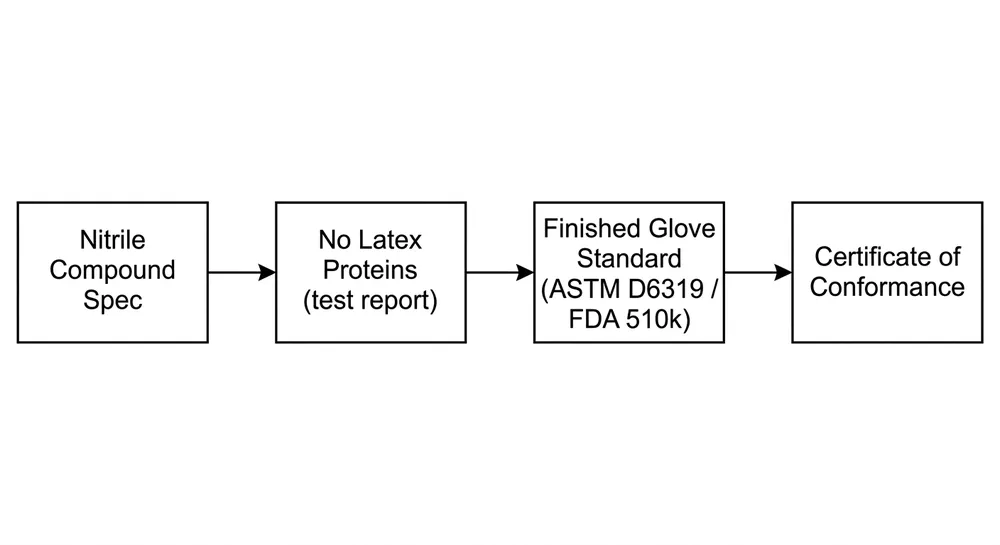
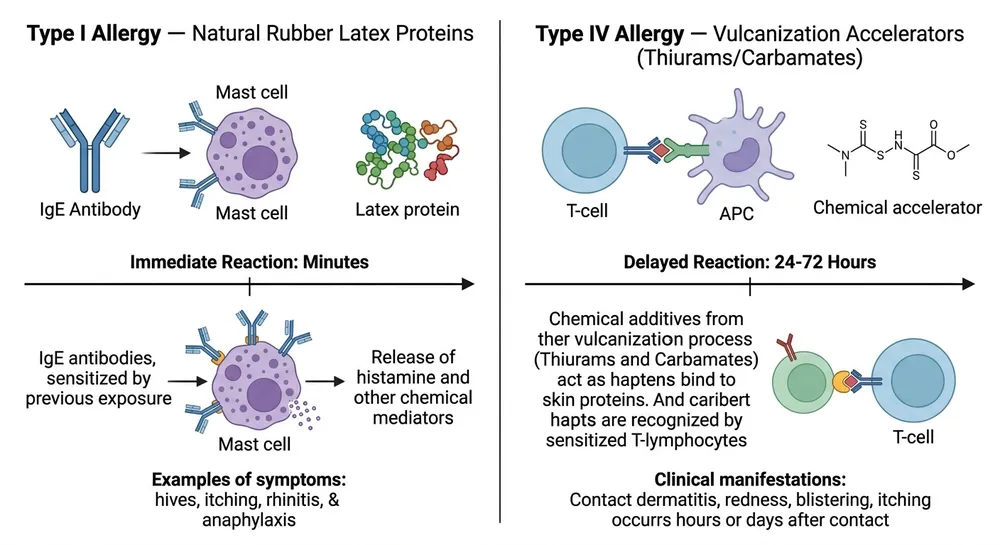
Type I — IgE-mediated latex allergy is an immediate hypersensitivity reaction to natural rubber latex proteins (primarily Hev b proteins). Symptoms appear within minutes: urticaria, rhinitis, in severe cases anaphylaxis. The affected population is workers with documented latex sensitization — historically a significant concern in healthcare settings before nitrile became the dominant glove material. Because all nitrile gloves contain zero natural rubber latex, any nitrile glove eliminates this risk. No special formulation required.
Type IV — T-cell mediated contact dermatitis is a delayed hypersensitivity reaction, typically appearing 24–72 hours after exposure. The trigger is not latex protein — it's the residual chemical accelerators left in the glove after vulcanization. The most common culprits are thiurams (thiuram disulfide, tetramethylthiuram) and carbamates (zinc diethyldithiocarbamate), which are standard components in conventional nitrile cure systems. A worker wearing standard nitrile gloves every day can develop progressive contact dermatitis from accelerator residue even though the gloves are fully latex-free.
This is the reaction that standard "latex-free" labeling does nothing to address — and it's the one that's increasingly showing up in occupational health reports, EU tender requirements, and hospital procurement specifications.
—
What "Latex-Free" Actually Covers — and What It Doesn't
When a nitrile glove is labeled latex-free, the claim is accurate and verifiable. Nitrile (acrylonitrile-butadiene rubber) is synthesized without natural rubber latex. EN 455-3 testing for residual latex proteins will return non-detectable results. FDA 510(k) clearance for exam gloves requires latex protein content disclosure. These are real, testable standards.
What latex-free labeling does not address:
- The presence or absence of thiuram accelerators
- The presence or absence of carbamate accelerators
- The total extractable chemical content beyond latex proteins
- Compliance with EN ISO 21420, which covers chemical sensitizers more broadly
A supplier who lists "latex-free, powder-free" on a spec sheet without disclosing accelerator content is giving you half the allergy picture. That's not necessarily deceptive — most standard nitrile gloves are manufactured with conventional accelerator cure systems, and for the majority of end users, this is fine. The issue arises when you're supplying into environments where Type IV sensitivity is a known or probable risk: surgical and exam glove use in healthcare, dental assistant roles with prolonged daily wear, food processing lines with high-frequency glove changes.
For those markets, "latex-free" is a necessary condition, not a sufficient one.
—
What "Accelerator-Free" Actually Requires at the Manufacturing Level
This is where the specification gets technically meaningful — and where supplier claims need scrutiny.
Conventional nitrile vulcanization uses sulfur-based accelerators (thiurams, carbamates, dithiocarbamates) to cross-link the polymer chains during the dipping and curing process. These accelerators dramatically speed up cure time and improve glove mechanical properties. The trade-off is residual accelerator content in the finished glove — typically in the range of parts per million, but sufficient to trigger Type IV sensitization in susceptible individuals with repeated exposure.
Accelerator-free nitrile gloves eliminate this by using an alternative cure chemistry. The two main approaches are:
Sulfur-donor cure systems — replace conventional accelerators with sulfur-donor compounds that provide cross-linking without leaving thiuram or carbamate residues. This is the approach we use on our dedicated accelerator-free line at glovemfg. The cure chemistry is more demanding to control — temperature uniformity across the dipping line matters more, and compound viscosity needs tighter management — but the result is a glove that tests non-detectable for the accelerators covered under EN ISO 21420 and European hospital tender specifications.
Peroxide cure systems — use organic peroxides as the cross-linking agent instead of sulfur chemistry entirely. Peroxide-cured nitrile tends to have slightly different mechanical properties (typically higher tensile strength, different elongation profile) and is used in some high-end surgical glove formulations.
The critical point for procurement: "accelerator-free" is a manufacturing claim that requires documentation. A supplier cannot make a standard nitrile glove accelerator-free by washing it or adding a coating. The compound formulation and cure system must be different from the ground up. If a supplier offers you an accelerator-free SKU but cannot provide test reports showing non-detectable thiuram and carbamate levels, the claim is unverifiable.
(We run our accelerator-free compound on a physically separate line — not just a different batch on the same line. Cross-contamination from conventional accelerator residue in the dipping tanks is a real risk if you're sharing equipment, and it's something worth asking about explicitly when you evaluate suppliers.)
—
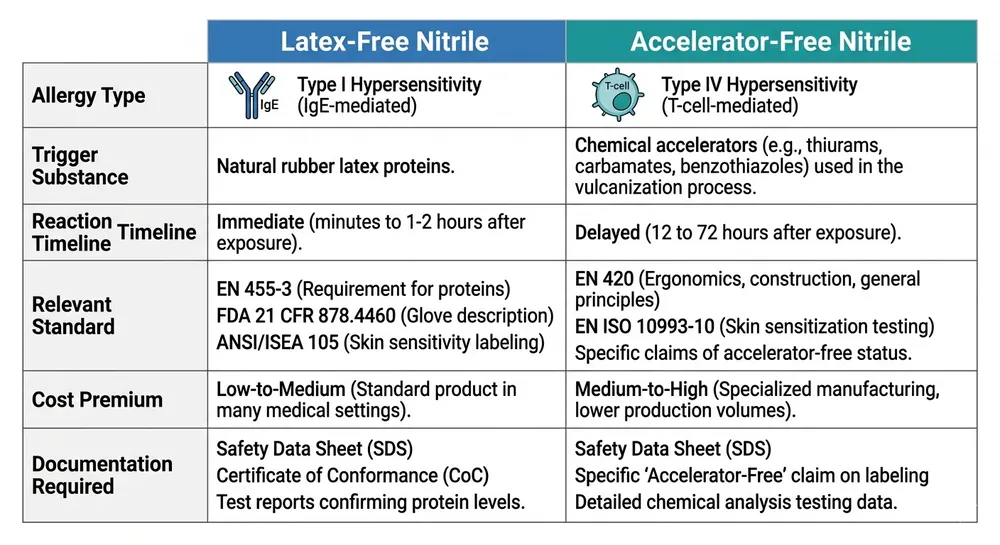
Side-by-Side: Latex-Free vs Accelerator-Free Nitrile
| Dimension | Latex-Free Nitrile (Standard) | Accelerator-Free Nitrile |
|---|---|---|
| Allergy mechanism addressed | Type I (IgE-mediated, latex proteins) | Type IV (T-cell mediated, chemical accelerators) |
| Trigger substance eliminated | Natural rubber latex proteins | Thiurams, carbamates, dithiocarbamates |
| Reaction timeline | Immediate (minutes) | Delayed (24–72 hours) |
| Relevant standard | EN 455-3 (latex protein content) | EN ISO 21420, EN 455-3 combined |
| Applicable to all nitrile? | Yes — all nitrile is latex-free by definition | No — requires specific accelerator-free formulation |
| Cost premium vs standard nitrile | None (baseline) | Moderate — typically 15–30% over standard nitrile |
| Availability at container scale | Universal | Limited to manufacturers with dedicated lines |
| Documentation to request | FDA 510(k) or CE/EN 455 certificate | EN ISO 21420 test report + accelerator residue panel |
| European hospital tender compliance | Necessary but not sufficient | Increasingly required as standalone specification |
| Typical end-user populations | General medical, industrial, food-contact | Healthcare workers with documented Type IV sensitivity, surgical teams, dental |
—
The Hidden Compliance Cost: What Happens When You Specify the Wrong Grade
The downstream cost of a misspecified glove order isn't the price difference between SKUs. It's the cost of a mid-contract switch.
A hospital system that specifies standard latex-free nitrile for a 12-month supply contract, then discovers three months in that several staff members are developing contact dermatitis consistent with Type IV accelerator sensitivity, faces a specific set of problems: occupational health investigations, potential workers' compensation exposure, an emergency re-specification process, and the cost of sourcing accelerator-free gloves on short notice — typically at spot pricing rather than contract pricing, and often from a different supplier than the original contract holder.
We've seen this play out with European hospital accounts. The pattern is consistent: the initial tender specifies "latex-free nitrile exam gloves" without an accelerator-free clause, the contract runs for a quarter, dermatitis cases surface in the occupational health log, and the procurement team is suddenly looking for an accelerator-free alternative mid-year. The re-sourcing cost — including the premium for short-notice container orders and the administrative overhead of a mid-contract supplier change — typically exceeds the cost difference between standard and accelerator-free gloves over the full contract term.
The smarter approach, particularly for healthcare and dental supply contracts, is to assess the end-user risk profile before the tender goes out. If the facility has any documented history of contact dermatitis in glove-wearing staff, or if the contract covers surgical or high-frequency exam use, the accelerator-free specification should be the default, not an upgrade.
For industrial and food-contact applications where glove wear is intermittent and Type IV sensitivity history is not documented, standard latex-free nitrile is the correct specification — and paying the accelerator-free premium is unnecessary cost.
—
Procurement Decision Guide: Matching Specification to End-User Risk
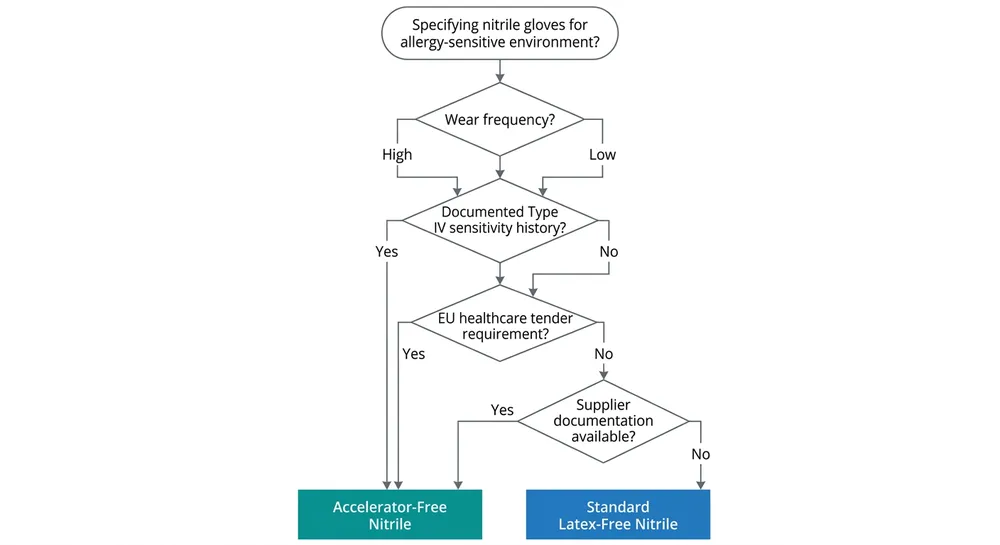
The decision isn't complicated once you have the right information. Here's how to work through it.
Step 1: Identify the end-user wear pattern. High-frequency, prolonged daily wear (surgical teams, dental assistants, exam glove use in clinical settings) carries higher Type IV sensitization risk than intermittent industrial use. The longer and more frequent the contact, the more relevant accelerator-free specification becomes.
Step 2: Check for documented sensitivity history. If the facility or workforce has any occupational health records of contact dermatitis attributed to glove use, accelerator-free is the correct specification regardless of wear frequency. This is not a precautionary upgrade — it's the clinically indicated grade.
Step 3: Assess the regulatory environment. European hospital tenders increasingly include explicit accelerator-free requirements, often citing EN ISO 21420 or referencing thiuram-free and carbamate-free as tender conditions. If you're supplying into EU healthcare procurement, check the tender language carefully — "latex-free" alone may not satisfy the allergy-safe specification.
Step 4: Evaluate your supplier's documentation capability. Accelerator-free claims require test reports. Specifically: an extractable accelerator panel showing non-detectable thiuram and carbamate levels, ideally from an accredited third-party lab. If your supplier cannot produce this documentation, the claim is unverifiable and the specification is commercially meaningless.
—
Application Scenarios: Which Specification Wins
Scenario 1: European Hospital Exam Glove Tender
A medical distributor is bidding on a 12-month supply contract for a regional hospital network in Germany. The tender specification references EN 455 compliance and includes a clause requiring gloves suitable for staff with documented contact dermatitis history.
Winner: Accelerator-free nitrile. The tender language effectively requires it. Standard latex-free nitrile satisfies EN 455-3 for latex protein content but does not address the accelerator sensitivity clause. Bidding standard nitrile risks disqualification or mid-contract replacement. The 15–30% cost premium over standard nitrile is absorbed into the contract pricing — and is far less than the cost of a mid-contract re-specification.
Scenario 2: US Industrial Safety Distributor, General Manufacturing Accounts
A safety products distributor supplies nitrile exam and industrial gloves to manufacturing facilities across the Midwest. End users are machine operators and assembly workers with no documented glove allergy history. Glove use is task-specific, not continuous daily wear.
Winner: Standard latex-free nitrile. The Type IV sensitization risk is low given intermittent use and no documented sensitivity history. The accelerator-free premium adds cost without addressing a documented risk. Standard latex-free nitrile with AQL 1.5 inspection and FDA 510(k) clearance is the correct specification for this account profile. Paying the accelerator-free premium here is margin erosion without clinical justification.
Scenario 3: Dental Supply Distributor, North American Market
A dental supply company is building a private-label nitrile glove line for dental practices. Dental assistants wear gloves for 6–8 hours daily. The company's existing accounts have flagged occasional staff complaints about hand irritation after extended wear.
Winner: Accelerator-free nitrile. The wear pattern (prolonged daily contact) and the existing irritation complaints are both indicators of probable Type IV sensitization risk. Switching to accelerator-free eliminates the most likely chemical trigger. For a private-label program, the accelerator-free specification also becomes a differentiator — dental practices are increasingly aware of this issue, and a supplier who can document accelerator-free compliance has a stronger value proposition than one who can only offer "latex-free."
—
Verifying Accelerator-Free Claims Before Your Container Ships
This is where most procurement processes fall short. The claim is easy to make; the documentation is what separates a verifiable specification from a marketing label.
What to request from any supplier claiming accelerator-free:
- Extractable accelerator test report — specifically covering thiuram disulfide, tetramethylthiuram monosulfide (TMTM), tetramethylthiuram disulfide (TMTD), zinc diethyldithiocarbamate (ZDEC), and zinc dibutyldithiocarbamate (ZDBC). Results should show non-detectable or below-threshold levels. Ask for the lab name and accreditation status.
- EN ISO 21420 compliance documentation — this standard covers chemical sensitizers in protective gloves and is the relevant European framework for accelerator-free claims in PPE procurement.
- Manufacturing process confirmation — ask whether the accelerator-free SKU is produced on a dedicated line or shares equipment with standard nitrile production. Shared equipment creates cross-contamination risk that can invalidate the accelerator-free claim even if the compound formulation is correct.
- Batch-level traceability — for healthcare contracts, request that the accelerator-free test report corresponds to the specific production batch in your order, not a generic product certification from a previous run.
At glovemfg, our accelerator-free Latex Free Blue Nitrile Gloves are produced on a dedicated line using a sulfur-donor cure system. The in-house compounding lab formulates the accelerator-free compound separately from our standard nitrile batches — this is what makes it possible to offer both grades from the same facility without subcontracting or relying on a third-party compounder. Test reports covering the accelerator residue panel are available with each order. If you're evaluating suppliers for an accelerator-free tender requirement, that documentation package is what you should be asking for as a baseline.
For a broader look at how allergy compliance intersects with blue nitrile glove specification, see our guide on latex-free blue nitrile gloves and allergy compliance.
—
What the Certifications Actually Cover
Certification language in glove procurement is frequently misread. Here's what the relevant standards actually address:
EN 455-3 — covers residual powder and chemical content in medical gloves, including latex protein levels. This is the standard that confirms a glove is latex-free in the clinical sense. It does not test for or set limits on accelerator content.
EN ISO 21420 — the European standard for protective gloves, covering chemical sensitizers including accelerators. This is the relevant standard for accelerator-free claims in PPE procurement. A glove that complies with EN ISO 21420 for chemical sensitizers has been tested against the accelerator compounds most associated with Type IV contact dermatitis.
ASTM D6319 — the US standard for nitrile exam gloves, covering physical performance (tensile strength, elongation, puncture resistance) and latex protein content. Like EN 455-3, it does not specifically address accelerator content. FDA 510(k) clearance references ASTM D6319 for exam gloves.
CE marking under EU MDR — medical device regulation compliance for exam and surgical gloves sold in Europe. CE marking requires EN 455 compliance but does not automatically require accelerator-free formulation unless the product is specifically labeled and tested as such.
The practical implication: a glove can carry CE marking, EN 455 compliance, and FDA 510(k) clearance and still contain conventional accelerator levels. These certifications confirm the glove is safe for general medical use — they don't confirm it's suitable for workers with documented Type IV sensitivity unless the accelerator-free specification is explicitly tested and documented separately.
(This is the gap that European hospital tenders are increasingly closing by adding explicit accelerator-free language to tender requirements — the existing certification framework doesn't automatically cover it.)
—
Cost and MOQ Reality at Container Scale
The accelerator-free premium is real but manageable at container scale. Here's how it typically breaks down.
Standard latex-free nitrile and accelerator-free nitrile share the same base polymer and most of the same manufacturing process. The cost difference comes from three sources: the alternative cure chemistry (sulfur-donor compounds cost more than conventional accelerators), tighter process control requirements on the dipping line, and lower production volume per SKU (accelerator-free runs are typically smaller than standard runs, which affects line efficiency).
At container scale — roughly 2,000 boxes per container for standard exam glove SKUs — the accelerator-free premium typically runs 15–30% over equivalent standard nitrile. The exact figure depends on glove thickness, size mix, and certification package required. For a healthcare distributor building a contract bid, this premium needs to be factored into the tender pricing from the start, not discovered after award.
MOQ for accelerator-free SKUs is generally higher than for standard nitrile because the dedicated line run needs to justify the compound changeover. At glovemfg, our standard MOQ for Blue Nitrile Gloves is one container (approximately 2,000 boxes) for standard SKUs. Accelerator-free SKUs follow the same container-scale MOQ — we don't require larger minimums for the accelerator-free grade, which is not always the case with manufacturers who subcontract their accelerator-free production.
For distributors building a mixed portfolio — standard nitrile for industrial accounts, accelerator-free for healthcare and dental — sourcing both grades from the same manufacturer simplifies the supplier relationship, consolidates the certification documentation, and reduces the logistics overhead of managing two separate supply chains.
—
Sourcing Checklist: What to Verify Before Placing an Accelerator-Free Order
Use this as your pre-order verification framework when evaluating suppliers for accelerator-free nitrile gloves:
- [ ] Compound formulation confirmed — supplier has confirmed the cure system (sulfur-donor or peroxide) and can describe why it eliminates conventional accelerator residues
- [ ] Dedicated production line — accelerator-free SKUs are not produced on shared equipment with standard nitrile
- [ ] Extractable accelerator test report available — covers thiurams (TMTM, TMTD) and carbamates (ZDEC, ZDBC) at minimum; results show non-detectable or below-threshold levels
- [ ] EN ISO 21420 compliance documentation — for European procurement; confirm the specific test scope matches your tender requirements
- [ ] Batch traceability — test reports can be linked to specific production batches, not just product-level certifications
- [ ] CE marking scope — confirm whether CE marking covers the accelerator-free claim specifically or only general EN 455 compliance
- [ ] MOQ and lead time confirmed — accelerator-free runs may have different scheduling than standard production; confirm before committing to a contract timeline
- [ ] Sample order available — request a pre-production sample with accompanying test report before committing to container volume
For a detailed guide on the accelerator-free medical nitrile gloves specification and what the manufacturing process looks like from the inside, that resource covers the formulation and QC side in more depth.
—
The Specification Decision, Summarized
Latex-free nitrile is the correct baseline for any application where natural rubber latex allergy is a concern — which, in practice, means all nitrile gloves already satisfy this requirement.
Accelerator-free nitrile is the correct specification when:
- End users have documented Type IV contact dermatitis history attributed to glove use
- The application involves prolonged daily wear (surgical, dental, high-frequency clinical exam use)
- The procurement is for European healthcare tenders that explicitly require accelerator-free or EN ISO 21420 compliance
- You're building a private-label program for healthcare or dental markets where allergy-safe differentiation has commercial value
The two specifications are not interchangeable, and "latex-free" does not imply "accelerator-free." If your current supplier's spec sheet lists latex-free without disclosing accelerator content, that's the first question to ask before your next container order.
If you're sourcing for a market where both grades are needed — standard nitrile for industrial accounts, accelerator-free for healthcare — request a quote with your target market, annual volume, and size mix. We'll confirm the correct SKU, the certification package that matches your tender or compliance requirements, and container pricing for both grades.