The first container usually looks fine. The problem shows up in the third or fourth shipment — gloves that are thinner than the sample, pinhole failures at the destination port, or a thickness measurement that passes your incoming check but fails your customer's incoming check because they're using a different sampling plan. By then you've already committed to a supplier, your customers are already stocked, and switching mid-year costs you more than the original price difference ever would have.
Spec inconsistency in bulk nitrile glove sourcing isn't a quality accident. It's a process control problem that starts upstream — in how the manufacturer manages compound formulation, dipping line parameters, and outgoing inspection. If you don't know what to look for before you place the order, you'll find out the hard way after it ships.
This guide walks through the sourcing process the way we see it from the factory side: what the real failure points are, what questions to ask, and what documentation to require before you commit to volume.
Where the Spec Drift Actually Starts
Most buyers assume thickness variation is a dipping problem. It usually isn't — or at least, it doesn't start there.
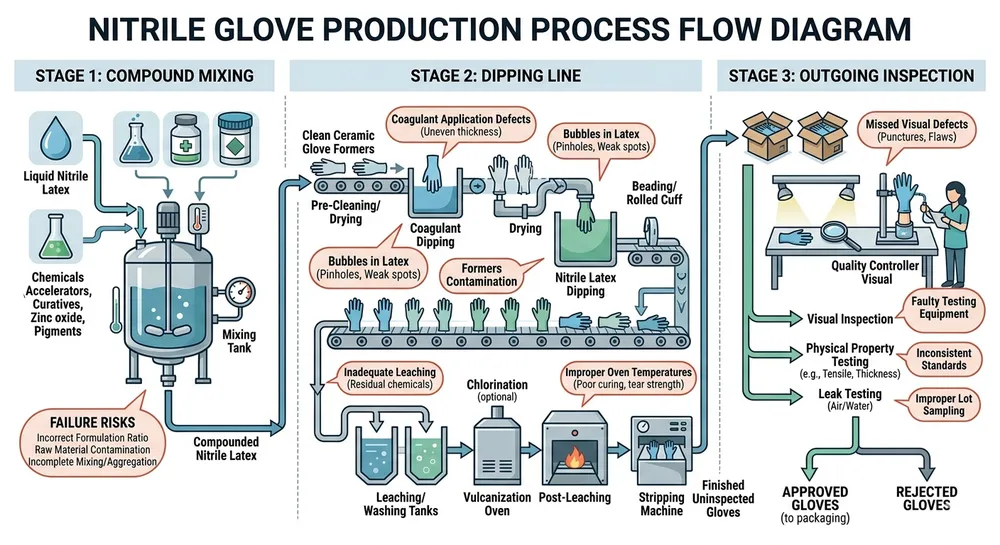
The real origin is compound consistency. Nitrile compound is a formulated mixture: acrylonitrile-butadiene polymer, vulcanizing agents, stabilizers, and in standard gloves, accelerators. If the factory buys pre-mixed compound from a third-party supplier, their batch-to-batch viscosity can shift by 5–10% without triggering any internal alarm. That viscosity shift changes how much compound adheres to the ceramic former on each dip pass, which changes wall thickness, which changes tensile strength and elongation results — all without anyone adjusting a single line parameter.
Factories that mix their own compound in-house can catch and correct this before it reaches the line. Factories that buy pre-mixed compound are dependent on their supplier's consistency, and many of them don't test incoming compound viscosity against a tight enough tolerance to catch the drift before it becomes a production problem.
The second failure point is line parameter drift. Automated dipping lines hold spec well when they're properly maintained and monitored — bath temperature, dipping speed, and compound viscosity need to stay within defined ranges throughout a production run. On a well-run line, bath temperature is held to ±1°C and viscosity is checked continuously. On a poorly maintained line, temperature can drift 3–5°C over a shift without triggering a shutdown, and that's enough to produce measurable thickness variation across a single lot.
The third failure point is inspection sampling. AQL 1.5 and AQL 2.5 are not the same standard. A factory running AQL 2.5 outgoing inspection will pass lots that would fail under AQL 1.5 — the acceptable defect rate is higher, the sample sizes are smaller, and the statistical protection for your downstream customers is weaker. If your supplier's quality documentation says "AQL inspection" without specifying the level, ask. The answer tells you a lot about where they've set their acceptable failure threshold.
Step 1: Define Your Spec Before You Ask for a Price
The most common sourcing mistake is leading with price. When you ask for a price before you've defined a specification, you get a quote for whatever the factory's standard SKU happens to be — and their standard SKU may not match what your customers actually need.
Before you contact any supplier, lock down these parameters:
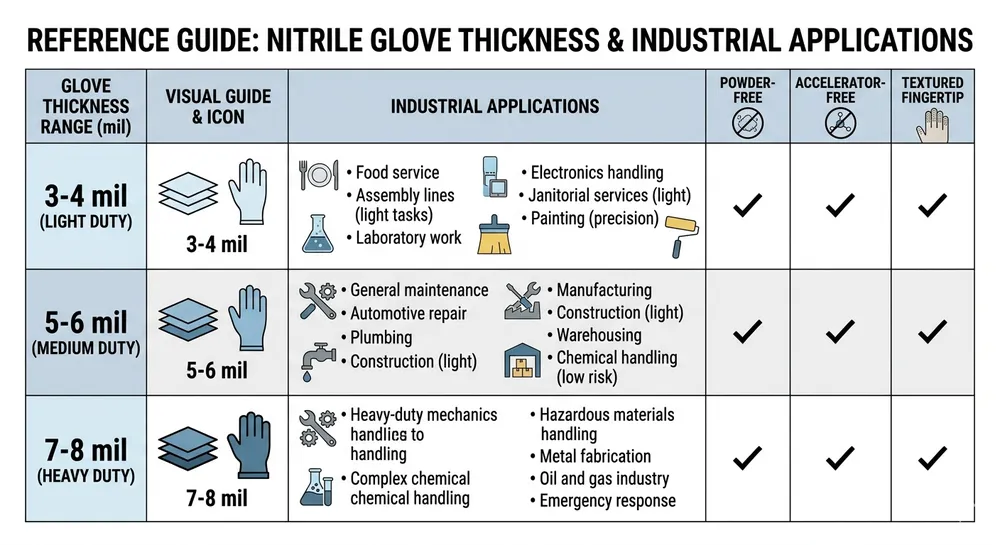
Thickness (mil or mm): Industrial nitrile gloves run from 3 mil (0.076mm) for light-duty assembly work up to 8 mil (0.203mm) for chemical handling. The right spec depends on the application. For general industrial use — assembly, light chemical splash, food processing — 4–5 mil (0.10–0.13mm) is the working range. For chemical handling with moderate exposure risk, 6–8 mil. Don't let a supplier talk you into a thinner glove because it's cheaper; the cost difference between 4 mil and 6 mil is small, and the liability difference if a glove fails in a chemical handling environment is not.
Powder-free vs. powdered: For most industrial applications today, powder-free is the correct spec. Powdered gloves are restricted or banned in medical applications in most export markets, and many industrial buyers have moved to powder-free to avoid contamination issues in food processing and electronics assembly. If your customers haven't specified, default to powder-free.
Accelerator-free: Standard nitrile uses thiuram and carbamate accelerators in vulcanization. These are the primary cause of Type IV allergic reactions. If you're supplying to healthcare-adjacent industrial environments — dental labs, pharmaceutical manufacturing, medical device assembly — accelerator-free is worth specifying. Not every factory can produce it; it requires a different cure chemistry and a dedicated line to avoid cross-contamination.
Textured vs. smooth: Fingertip texture improves grip on wet or oily surfaces. For automotive, metalworking, and wet-process industrial applications, textured fingertip is the right spec. For clean-room or electronics assembly, smooth is often preferred to avoid particle generation.
Certification requirements: If your customers operate in regulated procurement environments, you need to know which certifications are required before you qualify a supplier. EN 455 for European medical/industrial procurement, ASTM D6319 for North American markets, FDA 510(k) if your customers include US healthcare facilities. A supplier who holds these certifications has already passed third-party audits against defined performance standards — that's a meaningful baseline, not just a marketing claim.
Step 2: Qualify the Manufacturer, Not Just the Sample
A sample tells you what the factory can produce when they're trying. A qualification process tells you what they produce when they're not.
These are the questions that separate a consistent supplier from one who will drift on you by the third container:
Do they mix their own compound or buy pre-mixed? In-house compounding means the factory controls the formulation variables directly. They can adjust viscosity, cure profile, and chemical composition as a system. Pre-mixed compound means they're dependent on a supplier's batch consistency — and if that supplier has a bad batch, your order absorbs the variation. Ask directly. If they can't answer clearly, that's an answer.
What is their dipping line automation level? Fully automated lines with controlled conveyor speed, bath temperature monitoring, and viscosity checks hold tighter tolerances than semi-manual or operator-dependent lines. Ask for a production floor photo or video. You can see the difference between a modern automated line and an older semi-manual setup — the former has consistent former spacing, enclosed bath systems, and visible instrumentation. The latter has more operator touchpoints, which means more variation sources.
What AQL level do they run outgoing inspection at? AQL 1.5 is the tighter standard. AQL 2.5 is more common and more permissive. If a factory runs AQL 2.5, that's not automatically disqualifying — but you need to know, because it affects how you set your own incoming inspection plan. (We run 100% AQL 1.5 outgoing inspection across all production lots. That's a deliberate choice — it means our outgoing rejection rate is higher than a factory running AQL 2.5, but it also means fewer surprises at your destination port.)
Do they have online pinhole detection? Pinhole detection should run on every glove, not on a sampled basis. Electrical leak testing — where each glove passes through a charged field before stripping — catches pinholes that visual inspection misses. Ask whether pinhole detection is inline (every glove, automated) or offline (sampled, manual). Inline is the correct answer for any supplier you're considering for medical-adjacent or chemical handling applications.
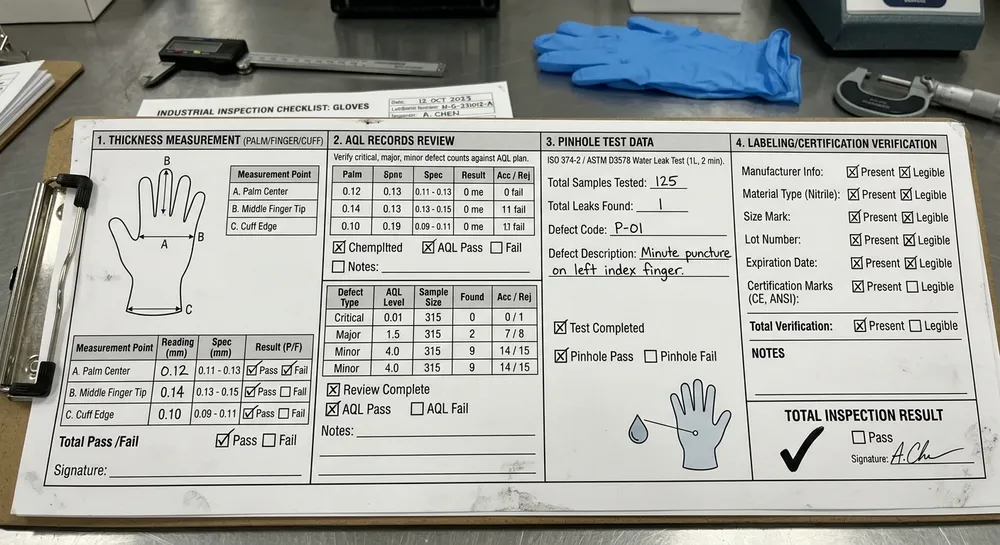
Can they provide batch-level test reports, not just lot-level certificates? A certificate of conformance tells you the lot passed. A batch-level test report tells you the actual measured values — thickness at palm, finger, and cuff; tensile strength; elongation at break; AQL defect count. The difference matters when you're trying to understand whether a supplier is consistently hitting the middle of the spec range or consistently skating the edge of it.
Step 3: Structure the Trial Order to Expose Variation
Don't test a supplier with a single sample shipment. Test them with a structured trial that's designed to surface the variation they'd rather you didn't see.
Order across two production runs, not one. Ask for your trial order to be split across two separate production runs — even if the total quantity is small. Spec consistency within a single run is easy. Consistency across runs, where the line may have been cleaned, the compound batch may have changed, and different shift operators may be involved, is the real test. If the supplier pushes back on this, that's worth noting.
Specify the test reports you want with each shipment. Before the trial order ships, tell the supplier exactly what documentation you need: thickness measurements at three points (palm, finger, cuff), tensile strength and elongation results, AQL inspection records with defect counts, and pinhole test pass/fail data. If they can't provide this documentation routinely, they're not running the process controls that generate it.
Run your own incoming inspection against the same parameters. When the trial shipment arrives, don't just do a visual check. Pull samples and measure thickness at the same three points the factory measured. Compare your results to their test report. A well-controlled factory's outgoing measurements and your incoming measurements should be close — within normal sampling variation. A significant gap between their reported values and your measured values is a red flag that their inspection process isn't as rigorous as their documentation suggests.
Check the packaging and labeling against your spec. This sounds basic, but labeling errors — wrong size marking, wrong lot number, missing certification references — are a common source of downstream problems for distributors. If the factory can't get the labeling right on a trial order when they know you're evaluating them, they won't get it right on a routine reorder.
Step 4: Lock the Spec in the Purchase Order, Not Just the Sample
The sample approval process is where most buyers think the spec is locked. It isn't — not unless the spec is written into the purchase order in measurable terms.
A purchase order that says "nitrile examination gloves, 4 mil, powder-free, blue" is not a locked spec. It's a description. A locked spec includes:
- Thickness range: e.g., 0.10mm ± 0.02mm at palm, 0.08mm ± 0.02mm at fingertip
- Tensile strength: minimum value in MPa, per ASTM D412 or EN 455-2
- Elongation at break: minimum percentage, per the same standard
- AQL level: specify AQL 1.5 or AQL 2.5 explicitly
- Pinhole test: specify inline electrical detection, 100% coverage
- Certification references: list the specific certifications required (EN 455, ASTM D6319, FDA 510(k))
- Labeling requirements: size, lot number, certification marks, language requirements for your target market
When these parameters are in the purchase order, you have a contractual basis for rejecting a shipment that doesn't meet them. When they're not, you're relying on the supplier's interpretation of "standard quality" — which may not match yours.
Some suppliers will push back on tight written specs, particularly on thickness tolerances. That pushback is informative. A factory that's confident in their process control will accept a written tolerance because they know they can hit it. A factory that's uncertain about their consistency will resist written tolerances because they know they can't guarantee them.
Step 5: Build Incoming Inspection Into Your Receiving Process
Even with a well-qualified supplier and a locked purchase order, incoming inspection is not optional for bulk industrial glove sourcing. The question is how to run it efficiently without creating a bottleneck at your receiving dock.
For standard reorders from a qualified supplier, a reduced incoming inspection plan is reasonable — pull samples from 10% of cartons, measure thickness at three points, check AQL against the supplier's outgoing report. If your results match theirs within normal sampling variation, release the lot. If there's a gap, expand the inspection before releasing.
For new suppliers, first orders, or any order where the supplier has changed their compound batch or production line configuration, run full incoming inspection against your purchase order spec. The cost of a thorough incoming inspection on a 2,000-box container is small compared to the cost of releasing a non-conforming lot to your customers.
Keep records. Incoming inspection data across multiple shipments from the same supplier is the most useful quality management tool you have. It tells you whether a supplier's consistency is improving, stable, or drifting — and it gives you the data to have a specific conversation with them when something changes.
Common Sourcing Mistakes That Show Up Later
These are the patterns we see most often when buyers come to us after a bad experience with a previous supplier:
Qualifying on price alone. The cheapest quote for a given spec description is usually cheap because something in the process is being cut — compound quality, inspection rigor, or line maintenance. The price difference between a well-controlled supplier and a poorly controlled one is typically 5–15% per box. The cost of a container rejection or a customer complaint is multiples of that.
Not specifying AQL level. Buyers who don't specify AQL level in their purchase order often discover after the fact that their supplier was running AQL 2.5 while they assumed AQL 1.5. The difference in acceptable defect rate between these two standards is significant enough to matter in practice.
Treating the first container as representative. Suppliers know which orders are being evaluated. The first container from a new buyer often gets extra attention — better compound batch selection, more careful inspection, faster production scheduling. The third container is the real test. Structure your qualification process to account for this.
Ignoring compound sourcing. Ask every potential supplier whether they mix their own compound or buy pre-mixed. This single question predicts more about long-term spec consistency than almost any other factor. Factories with in-house compounding labs have a structural advantage in consistency that pre-mixed buyers can't easily replicate.
Skipping the documentation review. Certifications are not self-certifications. EN 455, ASTM D6319, and FDA 510(k) all require third-party testing and, in the case of FDA 510(k), a formal clearance process. Ask for the actual certificate numbers and verify them. A supplier who can't provide verifiable certification documentation is a compliance risk for your customers.
What to Ask Before You Send an RFQ
Before you send a request for quotation to any supplier for industrial nitrile gloves in bulk, run through this checklist:
- [ ] Have you defined thickness, powder status, accelerator status, and texture in measurable terms?
- [ ] Do you know which certifications your customers require?
- [ ] Have you asked whether the factory mixes its own compound?
- [ ] Have you asked what AQL level they run outgoing inspection at?
- [ ] Have you asked whether pinhole detection is inline or sampled?
- [ ] Have you structured your trial order to span two production runs?
- [ ] Have you written your spec parameters into the purchase order, not just the sample approval?
- [ ] Do you have an incoming inspection plan for the first shipment?
If you can answer yes to all of these before you send the RFQ, you're sourcing from a position of information rather than assumption. That's the difference between a supplier relationship that holds spec for three years and one that surprises you at the worst possible time.
When you're ready to discuss specifications or request samples, send us your requirements — grade, size, application environment, and target market. We'll come back with a specific quote and the full certification documentation package for the SKUs that match your spec.