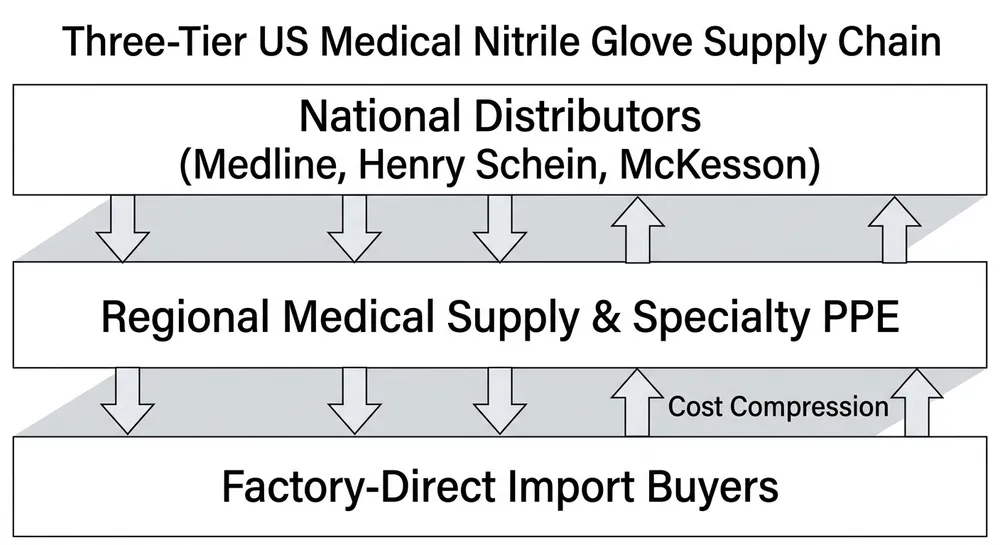
The US medical nitrile glove market runs on a layered supply chain. At the top are a handful of large national distributors who stock FDA-cleared product and can ship next-day to hospital systems. Below them sit regional medical supply companies, specialty PPE distributors, and a growing tier of direct-import buyers who have cut out the middle layer entirely. If you're building a procurement shortlist, understanding which tier each supplier operates in — and what that means for your unit cost, lead time, and compliance documentation — matters more than any ranked list.
This guide covers 10 real suppliers active in the US medical nitrile glove market, explains what each model is built for, and lays out the trade-off logic for when local stocking makes sense versus when factory-direct sourcing improves your landed cost and margin.
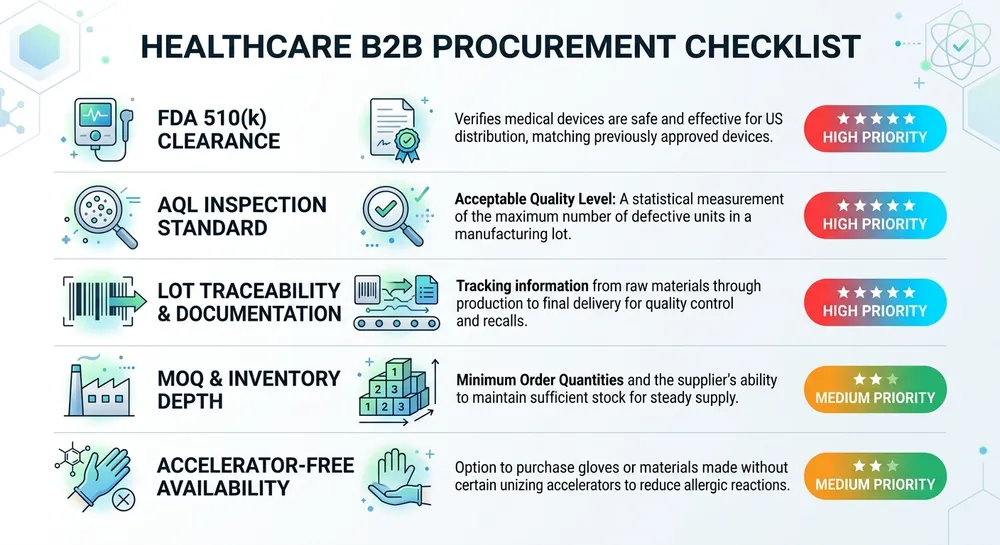
What Serious Procurement Teams Check Before Signing a Supplier
Before the shortlist, the criteria. Most hospital and clinic procurement teams are evaluating on five dimensions — and the weight of each shifts depending on whether you're buying for emergency stock or annual contract volume.
FDA 510(k) clearance is the baseline for exam-grade medical nitrile gloves in the US market. A supplier who can't produce a 510(k) number for the specific SKU you're buying is not a compliant source for clinical use. (We see this more than you'd expect — distributors listing "medical grade" product that traces back to an industrial-grade compound with no 510(k) on file.)
AQL inspection standard tells you how the gloves were tested before they left the factory. AQL 1.5 is the standard for medical-grade product under ASTM D6319. AQL 2.5 is common in industrial supply chains and gets misrepresented as equivalent. It isn't — at AQL 2.5, a lot with 2.5% defective units passes inspection. For clinical use, that's a meaningful difference.
Lot traceability and pre-shipment documentation matter when a container arrives and your receiving team needs to match the physical product to the compliance paperwork. Ask any supplier for a sample pre-shipment documentation package before you commit. If they can't produce one quickly, that's a signal about how their QC system is actually organized.
MOQ and inventory depth determine whether a supplier can serve your volume reliably across a full contract year — not just the first order. A distributor with shallow warehouse stock can fill a rush order but may not be able to hold spec consistency across 12 months of repeat orders.
Accelerator-free availability is increasingly relevant for hospital procurement, particularly in systems that have moved to Type IV allergy protocols. Not every supplier stocks accelerator-free nitrile as a standard SKU — some treat it as a special-order item with longer lead times and higher minimums.
The 10 Suppliers on the US Market Shortlist
These are real companies operating in the US medical nitrile glove supply chain. The list covers national distributors, regional medical supply companies, specialty PPE distributors, and one factory-direct manufacturer. Supplier type is noted for each — because the type determines the trade-offs, not the brand name.
—
1. Medline Industries — medline.com
Supplier type: National distributor / private-label manufacturer
Medline is the largest privately held medical products company in the US and one of the most significant players in hospital glove procurement. They distribute both branded and private-label nitrile gloves under their own Medline label, with FDA-cleared product across exam and procedure grades. Their supply chain infrastructure — regional distribution centers, EDI ordering, and GPO contract coverage — makes them the default choice for large hospital systems that prioritize procurement simplicity over unit cost optimization.
The trade-off: Medline's pricing reflects their distribution overhead and GPO margin structure. For high-volume buyers outside the GPO system, the unit economics are harder to justify against direct-import alternatives.
—
2. Henry Schein — henryschein.com
Supplier type: National distributor (dental, medical, and veterinary)
Henry Schein serves dental offices, physician practices, and ambulatory care facilities across the US. Their nitrile glove catalog covers exam-grade product from multiple manufacturers, with FDA 510(k) documentation available on request. For smaller clinic and practice accounts, Henry Schein's account management model and next-day delivery network are genuine advantages — the convenience premium is real, but so is the service.
Where they're less competitive: large-volume hospital procurement and private-label OEM. Henry Schein is a distributor, not a manufacturer, so customization options are limited to what their upstream suppliers offer.
—
3. McKesson Medical-Surgical — mckesson.com
Supplier type: National distributor / healthcare supply chain
McKesson is one of the three largest healthcare distributors in the US. Their medical-surgical division stocks nitrile exam gloves across multiple brands and grades, with deep integration into hospital GPO contracts (Vizient, Premier, HealthTrust). For IDN procurement teams working within an existing GPO framework, McKesson is often the path of least resistance.
The limitation is the same as any large distributor: you're buying through a supply chain layer, not from the source. Compliance documentation, lot traceability, and custom specification requests all route through McKesson's supplier relationships rather than directly to the factory.
—
4. Owens & Minor — owens-minor.com
Supplier type: National distributor / healthcare logistics
Owens & Minor operates one of the largest healthcare distribution networks in the US, with a focus on acute care and surgical supply. Their nitrile glove portfolio includes both commodity exam gloves and surgical-grade product. For hospital systems that have consolidated their supply chain through Owens & Minor's Byram or Halyard-branded programs, glove procurement often runs through existing account structures.
(Owens & Minor acquired Halyard Health's S&IP division in 2018 — so some product lines that buyers know under the Halyard name now route through Owens & Minor's distribution infrastructure.)
—
5. Cardinal Health — cardinalhealth.com
Supplier type: National distributor / private-label
Cardinal Health is the third major national healthcare distributor alongside McKesson and Owens & Minor. Their Flexal nitrile exam glove line is one of the more widely specified private-label products in US hospital procurement. FDA 510(k) cleared, available in powder-free formulations, and stocked across their distribution network.
For procurement teams already in the Cardinal Health ecosystem, Flexal is a straightforward specification. For buyers outside that ecosystem, the pricing and MOQ structure is less flexible than regional or direct-import alternatives.
—
6. Ansell Healthcare — ansell.com
Supplier type: Global PPE manufacturer with US distribution
Ansell is a global manufacturer — not a distributor — with manufacturing operations in Malaysia and other locations. Their Micro-Touch and TouchNTuff nitrile lines are widely specified in US hospital and laboratory settings. FDA 510(k) cleared, with AQL 1.5 inspection and documented lot traceability.
Ansell's position in the market is interesting: they're a manufacturer selling through distribution, which means you get manufacturer-level compliance documentation but distributor-level pricing. For buyers who want manufacturer accountability without managing a direct import relationship, Ansell is a credible middle option.
—
7. Kimberly-Clark Professional — kcprofessional.com
Supplier type: Global manufacturer / branded product
Kimberly-Clark's Purple Nitrile exam glove is one of the most recognized branded products in US clinical settings. FDA 510(k) cleared, with a distinctive formulation that has built strong end-user preference in surgical and procedure environments. For distributors serving accounts where brand preference drives the specification, Purple Nitrile commands a premium that holds.
The trade-off for procurement teams: brand premium is real and consistent. If your accounts are brand-agnostic and buying on compliance spec and unit cost, there are more cost-effective options at equivalent AQL and certification levels.
—
8. Dynarex Corporation — dynarex.com
Supplier type: Medical supply manufacturer / distributor hybrid
Dynarex is a US-based medical supply company that manufactures and distributes a broad range of disposable medical products, including nitrile exam gloves. Their product line covers powder-free nitrile in standard exam grades, FDA 510(k) cleared, with distribution through medical supply dealers and online channels. Dynarex occupies a mid-market position — more accessible MOQs than the large national distributors, with reasonable compliance documentation.
For regional medical supply distributors and smaller clinic accounts, Dynarex is a practical option where the large GPO-integrated distributors are oversized for the account.
—
9. Protective Industrial Products (PIP) — pipusa.com
Supplier type: Safety and PPE distributor
PIP is primarily an industrial safety distributor, but their medical-grade nitrile glove catalog has expanded significantly since 2020. They carry FDA-cleared exam gloves alongside their industrial PPE line, which makes them a practical single-source option for facilities that need both clinical and industrial nitrile product. Their distribution network covers industrial, healthcare, and government accounts.
The caveat: PIP's core competency is industrial PPE, not medical compliance. For hospital procurement teams with strict documentation requirements, verify the specific SKU's 510(k) status and AQL inspection records before specifying.
—
10. Glovemfg (Qingdao Glovemfg Medical Products Co., Ltd.) — glovemfg.com
Supplier type: Factory-direct manufacturer (China, exporting to US market)
We're on this list because we're a real option for US buyers who have outgrown the distributor model — and being transparent about what that means is more useful than pretending we're a domestic supplier.
We've been manufacturing nitrile gloves since 2007. The facility in Qingdao runs 8 fully automated dipping lines across 18,000 square meters, with annual output at 1.2 billion pieces. Our medical nitrile gloves carry FDA 510(k) clearance, CE marking, EN 455, and ASTM D6319 certification. AQL 1.5 inspection is standard — not an upgrade tier. We run 100% online pinhole detection on every line.
What makes the factory-direct model work at volume: when you're buying through a national distributor, you're paying for their warehousing, their account management infrastructure, and their margin. At container-scale repeat procurement — roughly 2,000 boxes per container as a starting point — the landed cost difference is significant enough to change the unit economics of your distribution business. We've seen US distributors move from GPO-sourced product to direct-import and recover 15–25% on landed cost, depending on the SKU and their existing freight relationships.
The honest trade-offs: lead time from order to container is longer than domestic stock. We're not the right answer for a 500-box emergency order that needs to ship tomorrow. We are the right answer for a buyer who knows their annual volume, wants to own their compliance documentation directly, and is ready to run a 12-month supply agreement instead of spot-buying through a distributor.
(We also support OEM private-label — if you're building your own brand on medical nitrile, we can run your label from the same lines that produce our FDA-cleared standard product. MOQ for OEM starts at one container.)
—
Where Local Distribution Earns Its Premium
Before making the case for factory-direct, it's worth being direct about when local distribution is the right answer — because it genuinely is, in specific scenarios.
Emergency and short-cycle replenishment: If a hospital system burns through stock faster than projected, or a clinic needs to cover a supply gap within 48 hours, a national distributor with regional warehouse coverage is the only viable option. No factory-direct relationship solves a same-week emergency.
GPO contract compliance: Many hospital systems are contractually obligated to source through GPO-approved suppliers. If your procurement is governed by a Vizient, Premier, or HealthTrust contract, the GPO framework determines your supplier options regardless of unit cost comparisons.
Low-volume accounts: For a small clinic buying 50–100 boxes per quarter, the economics of direct import don't work. The container MOQ, the freight management, and the customs documentation overhead are all sized for buyers operating at higher volume. A regional distributor is the right fit.
Brand-specified accounts: Some end-user accounts specify by brand — Kimberly-Clark Purple Nitrile, for example, has genuine end-user preference in certain surgical environments. If your downstream customer is specifying by brand, you're buying that brand regardless of the sourcing economics.
The local distribution premium is real and justified in these scenarios. The question is whether your procurement volume and contract structure put you in a position to capture the cost difference that factory-direct sourcing offers.
The Cost of the Distribution Layer at Scale
For buyers operating at container-scale volume — annual procurement in the range of 500,000 to several million pairs — the distribution layer has a measurable cost. It's not a criticism of distributors; it's the arithmetic of how supply chains work.
A national distributor sources product from manufacturers (often overseas), warehouses it domestically, manages their own compliance and logistics infrastructure, and sells it with a margin that covers all of that. That margin is typically 20–40% above the manufacturer's export price, depending on the product tier and the distributor's cost structure. For commodity exam gloves — a product where the compliance spec is standardized and the differentiation is thin — that margin is the primary variable in your landed cost.
At 1 million pairs per year, a 20% cost difference is a number that shows up clearly in your P&L. At 5 million pairs, it's a sourcing decision that determines whether your distribution business is competitive in your market.
The factory-direct model doesn't eliminate all overhead — you're taking on freight management, customs clearance, and pre-shipment inspection coordination. But for buyers with the volume and operational capacity to manage a direct import relationship, the economics are straightforward.
Matching Sourcing Route to Procurement Scenario
| Scenario | Recommended Sourcing Route |
|---|---|
| Emergency stock, 48-hour need | National distributor (Medline, McKesson, Cardinal Health) |
| GPO-contracted hospital system | GPO-approved distributor per contract terms |
| Small clinic, <200 boxes/quarter | Regional distributor or Dynarex |
| Brand-specified account (Purple Nitrile, etc.) | Branded manufacturer's distribution channel |
| Mid-size distributor, 500K+ pairs/year | Evaluate factory-direct; run landed cost comparison |
| Private-label OEM, building your own brand | Factory-direct manufacturer with OEM capability |
| Annual contract, 1M+ pairs, cost-sensitive | Factory-direct import; container-scale economics apply |
| Accelerator-free specification, EU or US hospital | Verify with manufacturer directly; not all distributors stock |
The decision isn't binary. Some buyers run a hybrid model: domestic distributor for emergency and spot orders, factory-direct for their base annual volume. That structure captures the convenience of local stock for exceptions while recovering the cost difference on the bulk of their procurement.
Verifying Compliance Before You Commit
One practical note on FDA 510(k) verification: the FDA's 510(k) database is publicly searchable at accessdata.fda.gov. Before finalizing any supplier for clinical use, search the specific product name or the manufacturer's name and confirm the clearance number matches the SKU you're buying.
We've seen situations where a distributor's catalog lists a product as "FDA cleared" but the clearance on file is for a different size, formulation, or even a different product entirely. The 510(k) number should be on the product packaging and traceable to a specific device listing. If a supplier can't provide that number on request, that's a compliance gap worth taking seriously before a container ships.
For EN 455 compliance in European markets, the same principle applies — the notified body certificate should reference the specific product and manufacturing site, not just the company name.
Factory-Direct as a Strategic Sourcing Layer
If your procurement volume puts you in the range where factory-direct economics make sense, the evaluation process is straightforward. Request a pre-shipment documentation package — this should include the 510(k) clearance number, AQL inspection report for the specific lot, material safety data, and the certificate of conformance. A manufacturer who can't produce this package quickly is not organized for compliant medical export.
For our part, we've been shipping to US healthcare distributors since our early years. The documentation package is standard, the AQL 1.5 inspection is built into the production process rather than bolted on at the end, and our in-house compounding lab means we can hold formulation consistency across a full year's worth of orders — not just the first sample shipment.
If you're evaluating a direct-import relationship for your medical nitrile gloves procurement, the most useful first step is a landed cost comparison against your current distributor pricing. We can provide FOB pricing, standard freight estimates for your destination port, and a sample documentation package so you can run that comparison with real numbers. Request a quote with your target volume, size mix, and any specification requirements — accelerator-free, specific thickness range, OEM label — and we'll respond with a detailed breakdown.