Most container-level sourcing problems with medical nitrile gloves don't start at the factory. They start at the specification stage, when a buyer assumes that "FDA-cleared" or "EN 455 certified" on a product sheet means the lot in their container was made to the same standard as the sample that earned the clearance. It often wasn't.
This guide walks through what FDA 510(k), EN 455, and AQL 1.5 actually require — and what they don't cover — so you can ask the right questions before you commit to a supplier, not after a container arrives at your warehouse with a compliance problem.
—
What FDA 510(k) Clearance Actually Covers — and What It Doesn't
FDA 510(k) clearance means the glove design and manufacturing process were reviewed and found substantially equivalent to a legally marketed predicate device. That review happens once, at the time of submission. It does not mean every production lot is tested by the FDA. It does not mean the factory's current output matches the cleared specification.
What 510(k) clearance tells you:
- The glove design (material, thickness range, intended use) was reviewed and accepted
- The manufacturer's quality system was evaluated at the time of clearance
- The product is legally marketable as a medical device in the US under that 510(k) number
What it doesn't tell you:
- Whether the current production lot meets the cleared thickness and physical property specs
- Whether the factory has changed its nitrile compound formulation since clearance
- Whether the AQL inspection standard applied to your specific shipment matches what was declared in the 510(k)
The 510(k) number is a starting point for verification, not a substitute for it. When we handle documentation for US distributors, the first thing we pull is the 510(k) summary — specifically the predicate device comparison and the performance testing data. If a supplier can't produce that summary on request, that's a flag worth taking seriously.
(We've seen cases where a factory holds a valid 510(k) but has since reformulated their compound to reduce cost. The clearance number stays the same. The glove doesn't.)
For medical nitrile gloves destined for US healthcare distribution, the 510(k) number should be verifiable on the FDA's 510(k) database. Cross-reference the applicant name, device description, and clearance date before accepting a product sheet at face value.

—
EN 455: Four Parts, One Standard — and Why Part 3 Is the One Buyers Miss
EN 455 is the European standard for single-use medical examination gloves. It has four parts, and most product sheets only mention the standard by name without specifying which parts were tested. That matters because each part covers a different performance dimension.
| EN 455 Part | What It Tests | Why It Matters for Your Order |
|---|---|---|
| Part 1 | Freedom from holes (AQL leak test) | Barrier integrity — the baseline |
| Part 2 | Physical properties (tensile strength, elongation, thickness) | Durability under clinical use conditions |
| Part 3 | Biological evaluation (biocompatibility, residual chemicals) | Accelerator content, protein levels, extractable chemicals |
| Part 4 | Shelf life determination | Packaging integrity and aging stability |
Part 3 is where most sourcing problems hide. It covers residual chemical levels — including thiuram and carbamate accelerators, which are the primary drivers of Type IV allergic contact dermatitis in healthcare workers. European hospital procurement increasingly requires accelerator-free gloves specifically because of EN 455-3 compliance pressure.
If you're supplying into European healthcare accounts or US hospital systems with sensitization policies, "EN 455 certified" on a spec sheet is not enough. You need the EN 455-3 test report, and you need to confirm whether the glove is accelerator-free or accelerator-reduced. Those are different things. Accelerator-reduced gloves still contain thiurams and carbamates — just at lower concentrations. Accelerator-free means the compound was formulated without them from the start.
We run an in-house compounding lab specifically because accelerator-free formulation requires control at the compound level, not just at the finished-glove testing stage. You can't remove accelerators from a standard compound after the fact — the chemistry doesn't work that way. The formulation decision happens before the dipping line runs.

—
AQL 1.5: What the Number Means and How It Gets Misapplied
AQL stands for Acceptable Quality Limit. AQL 1.5 for medical examination gloves means that in a statistically valid sample from a production lot, no more than 1.5% of units may be defective — specifically, no more than 1.5 pinholes per 100 gloves tested under the EN 455-1 or ASTM D6319 water-leak test protocol.
The number sounds tight. In practice, the rigor depends entirely on the sample size used and whether the inspection was performed on the production lot you're actually receiving.
Here's where buyers get caught:
Sample size matters. AQL 1.5 at inspection level II (the standard for medical devices under ISO 2859-1) requires a much larger sample than AQL 1.5 at inspection level I. A supplier can claim AQL 1.5 compliance while using a reduced sample size that statistically allows more defects through. Ask for the inspection level, not just the AQL number.
Pre-shipment vs. in-line inspection. AQL inspection can be performed in-line during production or as a pre-shipment check on finished goods. In-line inspection catches defects earlier and allows for lot rejection before packaging. Pre-shipment inspection on a finished lot is better than nothing, but it's a lagging indicator. We run online pinhole detection on all 8 dipping lines — every glove passes through the detection system before it reaches the packaging stage, not after.
Third-party vs. factory-issued CoA. A Certificate of Analysis issued by the factory is not the same as a third-party inspection report. For container-scale orders, a pre-shipment inspection by SGS, Bureau Veritas, or an equivalent third party gives you independent verification of the AQL result. Factory CoAs are useful for lot traceability, but they don't replace independent inspection for high-stakes shipments.
(The most common complaint we hear from buyers who've had problems with previous suppliers isn't that the gloves failed — it's that the CoA said they passed and the gloves still failed at destination. That gap almost always traces back to sample size or inspection timing.)
—
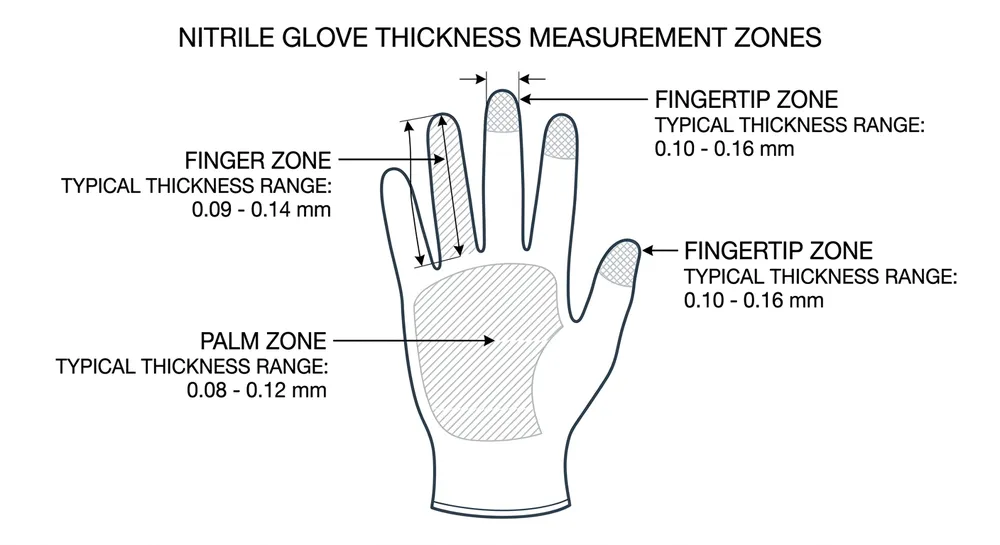
Thickness, Tensile Strength, and the Specs That Actually Predict Field Performance
Medical nitrile gloves for examination use typically run 0.08–0.12mm at the palm, with fingertip thickness slightly higher due to the dipping geometry. ASTM D6319 and EN 455-2 both set minimum tensile strength and elongation requirements, but the minimums are floors, not targets.
| Property | ASTM D6319 Minimum | EN 455-2 Minimum | Practical Range for Medical Use |
|---|---|---|---|
| Thickness (palm) | 0.05mm | Not specified | 0.08–0.12mm |
| Tensile strength (before aging) | 14 MPa | 6 N/cm² | 18–24 MPa typical |
| Elongation at break (before aging) | 500% | 500% | 550–650% typical |
| Tensile strength (after aging) | 14 MPa | 6 N/cm² | Should retain >80% of pre-aging value |
The aging test is the one that separates well-formulated compounds from cost-cut ones. Accelerated aging simulates shelf life — a glove that meets tensile minimums fresh but drops below spec after aging has a compound stability problem. That problem shows up at the end of the supply chain, when your customer opens a box from the back of their storage room.
We hold our compound formulation to 0.08–0.15mm thickness tolerance across the palm and fingertip zones. That tolerance is tighter than the ASTM minimum because thickness variation is the primary driver of AQL failures at destination — a glove that measures 0.10mm at the palm and 0.07mm at the fingertip will fail the water-leak test at the thin point, not the average.
Textured fingertip variants add a surface profile that affects grip under wet conditions. The texture is formed during the dipping process by controlling the coagulant application — it's not a post-process treatment. That matters because post-process texturing can create micro-tears in the film that compromise barrier integrity. If a supplier offers textured gloves, ask whether the texture is formed in-line or applied after dipping.
—
The Documentation You Should Request Before a Container Ships
Compliance documentation for medical nitrile gloves falls into two categories: device-level certification (issued once, covers the product design) and lot-level documentation (issued per production run, covers the specific goods in your container).
Most sourcing disputes trace back to buyers who verified device-level certification but didn't request lot-level documentation before payment.
Device-level documents to verify:
- FDA 510(k) clearance letter and summary (verify on FDA database)
- EN 455 Parts 1–4 test reports (issued by a notified body, not self-declared)
- ISO 9001:2015 certificate (verify issuing body and expiry date)
- ASTM D6319 test report if US market is the destination
Lot-level documents to request per shipment:
- Certificate of Analysis (CoA) with lot number, production date, AQL result, and inspection level
- Pre-shipment inspection report (third-party preferred for container-scale orders)
- Packing list with box count, glove count per box, and lot traceability
- For accelerator-free claims: EN 455-3 test report specific to the compound batch
One thing we do that most factories don't: we maintain lot traceability back to the compound batch. If a quality issue surfaces after delivery, we can pull the compound batch record, the in-line inspection data, and the pre-shipment CoA for that specific lot. That traceability is what makes a quality claim resolvable instead of a dispute.
If a supplier can't provide lot-level documentation before shipment, that's not a paperwork problem — it's a quality system problem.
—
Powder-Free, Accelerator-Free, and Latex-Free: Three Claims That Mean Different Things
These three terms appear together on product sheets often enough that buyers sometimes treat them as interchangeable. They're not.
Powder-free means the glove was manufactured without cornstarch or other powder lubricants. This is now the standard for medical examination gloves — powdered gloves were banned by the FDA for most medical uses in 2017. If a supplier is still offering powdered medical examination gloves for US distribution, that's a compliance problem, not a product option.
Latex-free means the glove contains no natural rubber latex. Nitrile gloves are inherently latex-free — nitrile is a synthetic rubber. The claim is accurate but redundant for nitrile. Where it matters is in documentation for hospital procurement systems that require explicit latex-free certification for allergy management protocols.
Accelerator-free is the claim that requires the most scrutiny. Nitrile gloves are vulcanized using chemical accelerators — typically thiurams, carbamates, or mercaptobenzothiazole compounds. These accelerators can remain as residuals in the finished glove and are the primary cause of Type IV contact dermatitis in healthcare workers. Accelerator-free formulation means the compound was developed using an alternative vulcanization chemistry that eliminates these residuals.
Not all suppliers who claim "accelerator-free" have actually reformulated their compound. Some reduce accelerator levels and call the result accelerator-free. The EN 455-3 test report is the verification document — it will show residual chemical levels. If a supplier claims accelerator-free but can't produce an EN 455-3 report with extractable chemical data, the claim is unverified.
We developed our accelerator-free compound in-house through our compounding lab. The formulation took longer to stabilize than a standard compound — alternative vulcanization chemistry affects cure time and tensile properties, and getting both right without accelerators required iteration. The EN 455-3 data is available for any buyer evaluating our accelerator-free SKU.
—
Sourcing at Container Scale: MOQ, Lead Time, and What "OEM" Actually Involves
For US healthcare distributors sourcing at container scale, the practical sourcing questions come down to minimum order quantities, lead time reliability, and what private-label OEM actually requires in terms of specification and documentation.
Our standard MOQ is one container — approximately 2,000 boxes for standard examination glove SKUs. That's roughly 200,000 pairs depending on box count configuration. For buyers new to direct factory sourcing, one container is a workable trial volume that lets you verify quality and documentation before committing to annual volume agreements.
OEM private-label for medical nitrile gloves involves more than printing your brand on the box. For US distribution, your private-label product needs to be covered by a 510(k) clearance — either your own or the manufacturer's, depending on your regulatory strategy. If you're using the manufacturer's 510(k), you need to confirm that your label configuration is covered under that clearance. If you're building toward your own 510(k), the manufacturer needs to be named in your quality agreement and capable of providing the manufacturing documentation the FDA requires.
(This is where a lot of first-time private-label buyers get surprised. The 510(k) question comes up at customs, not at the factory. Better to resolve it before the container ships.)
We support both paths — supplying under our existing FDA 510(k) for distributors who want to move quickly, and providing full manufacturing documentation for buyers building their own regulatory submissions. The documentation package for the latter includes compound batch records, in-process inspection data, and finished product test reports by lot.
For buyers evaluating us against other suppliers, the question worth asking any factory is: can you provide lot-level traceability documentation for the last three shipments you made to a US healthcare distributor? The answer tells you more about their quality system than any certification list.
—
Common Sourcing Mistakes That Show Up After the Container Arrives
These aren't hypothetical. They're the patterns we see when buyers come to us after a problem with a previous supplier.
Accepting a sample that doesn't represent production. Samples are often made on a dedicated run with tighter QC than standard production. The sample passes every test. The container doesn't. The fix is to request a pre-shipment inspection on the actual production lot, not just the sample.
Verifying the 510(k) number without checking the device description. A 510(k) number can be valid but cover a different glove configuration than what you're ordering. Check that the cleared device description matches the product you're buying — material, thickness range, intended use.
Treating AQL 1.5 as a pass/fail binary. AQL 1.5 is a statistical sampling standard. A lot that "passes" AQL 1.5 can still contain defective units — the standard defines an acceptable defect rate, not zero defects. For high-sensitivity applications, ask about the inspection level and sample size used.
Not specifying size distribution in the purchase order. Medical examination gloves are ordered in size runs — typically S/M/L/XL. If your PO doesn't specify the size distribution, the factory will ship the distribution that's most efficient for their production schedule, which may not match your market demand.
Assuming EN 455 certification covers the US market. EN 455 is a European standard. It's relevant for European distribution and useful as a quality benchmark, but it doesn't substitute for FDA 510(k) clearance for US medical device distribution. Both are needed if you're supplying into both markets.
—
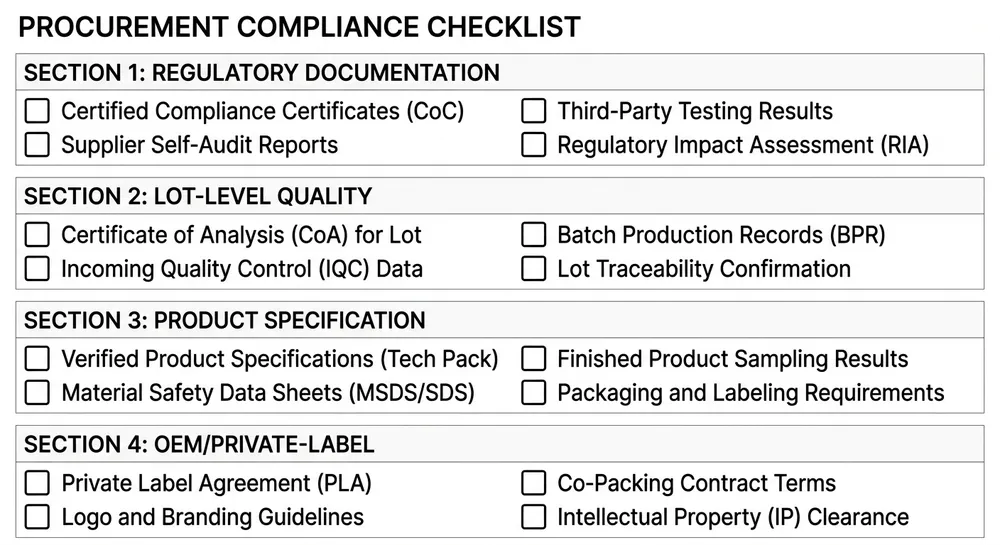
Evaluating a Medical Nitrile Glove Supplier: A Practical Checklist
Before committing to a container-scale order, these are the verification points worth working through systematically.
Regulatory documentation:
- [ ] FDA 510(k) number verified on FDA database, device description matches product
- [ ] EN 455 Parts 1–4 test reports available (notified body issued, not self-declared)
- [ ] ISO 9001:2015 certificate current and issuing body verifiable
- [ ] ASTM D6319 test report available for US-destined product
Lot-level quality:
- [ ] CoA format includes lot number, production date, AQL result, and inspection level
- [ ] Pre-shipment inspection by third party available or arrangeable
- [ ] Lot traceability to compound batch confirmed
Product specification:
- [ ] Thickness range specified and confirmed against 510(k) cleared range
- [ ] Accelerator-free claim supported by EN 455-3 extractable chemical data (if applicable)
- [ ] Powder-free confirmed (required for US medical use)
- [ ] Textured fingertip: in-line formation confirmed (not post-process)
OEM/private-label (if applicable):
- [ ] 510(k) coverage for your label configuration confirmed
- [ ] Quality agreement in place naming manufacturer
- [ ] Manufacturing documentation package scope agreed before order placement
—
FAQ: Medical Nitrile Glove Procurement for US Healthcare Distribution
What is the difference between FDA 510(k) clearance and FDA registration for nitrile gloves?
FDA registration means the manufacturing facility is registered with the FDA as a medical device manufacturer — it's a facility-level requirement. FDA 510(k) clearance is a product-level authorization that allows a specific glove design to be marketed as a medical device in the US. Both are required for legal US medical device distribution. A factory can be FDA-registered without having 510(k) clearance for a specific product, and a 510(k) clearance doesn't automatically mean the manufacturing facility is currently registered. Verify both independently.
Can I use EN 455-certified gloves for US hospital distribution without FDA 510(k) clearance?
No. EN 455 is a European standard and does not satisfy FDA requirements for medical device marketing in the US. Gloves sold as medical examination gloves in the US must have FDA 510(k) clearance. EN 455 certification is useful as a quality benchmark and required for European distribution, but it's not a substitute for 510(k) clearance in the US market.
What AQL level should I specify for medical examination gloves?
AQL 1.5 is the standard for medical examination gloves under both EN 455-1 and ASTM D6319. Some hospital procurement specifications require AQL 1.0 for higher-risk applications. When specifying AQL, also specify the inspection level (Level II is standard for medical devices under ISO 2859-1) and whether you require third-party pre-shipment inspection. AQL level alone, without inspection level and sample size, is an incomplete specification.
How do I verify that a supplier's accelerator-free claim is genuine?
Request the EN 455-3 test report for the specific compound batch used in your order. The report should show extractable chemical levels for thiurams, carbamates, and mercaptobenzothiazole compounds. A genuine accelerator-free glove will show non-detectable or negligible levels of these compounds. If the supplier can only provide a general product certification rather than a compound-specific test report, the claim is unverified for your specific lot.
What is a reasonable MOQ for a first container order of private-label medical nitrile gloves?
One container (approximately 2,000 boxes for standard examination gloves) is a workable first order for most distributors. Below that volume, the economics of private-label setup — label artwork, regulatory documentation, box configuration — typically don't justify the per-unit cost. For buyers new to direct factory sourcing, a single-container trial order with full pre-shipment inspection is a lower-risk way to verify quality and documentation before committing to annual volume.
What documents should I request before a container of medical nitrile gloves ships?
At minimum: the Certificate of Analysis for the production lot (with lot number, AQL result, and inspection level), the packing list with lot traceability, and confirmation that the 510(k) clearance covers the specific product configuration in your order. For container-scale orders, a third-party pre-shipment inspection report from SGS, Bureau Veritas, or equivalent adds independent verification. For accelerator-free products, the EN 455-3 test report for the compound batch used in your order.
—
If you're evaluating suppliers for a container-scale order of medical nitrile gloves and want to work through the specification and documentation requirements before committing, we're straightforward to work with. Send your grade requirements, size distribution, and destination market to our team and we'll put together a detailed quote with the full documentation package — Request a Quote.