Most US distributors who contact us about private label have already done some research. They know they want their brand on the box. What slows them down — sometimes by months — is not finding a factory. It's figuring out what they actually own in a private label arrangement, what compliance documentation they need to hold, and whether the factory they're talking to can deliver consistent product across a full year of orders, not just a sample shipment.
This article covers the mechanics: what private label medical nitrile gloves actually involve, how OEM and ODM differ in practice, what MOQ looks like at container scale, and what the FDA 510(k) and ASTM D6319 compliance picture means for your branded resale program.
What You Own in a Private Label Arrangement — and What the Factory Owns
The confusion here is common and worth clearing up before anything else.
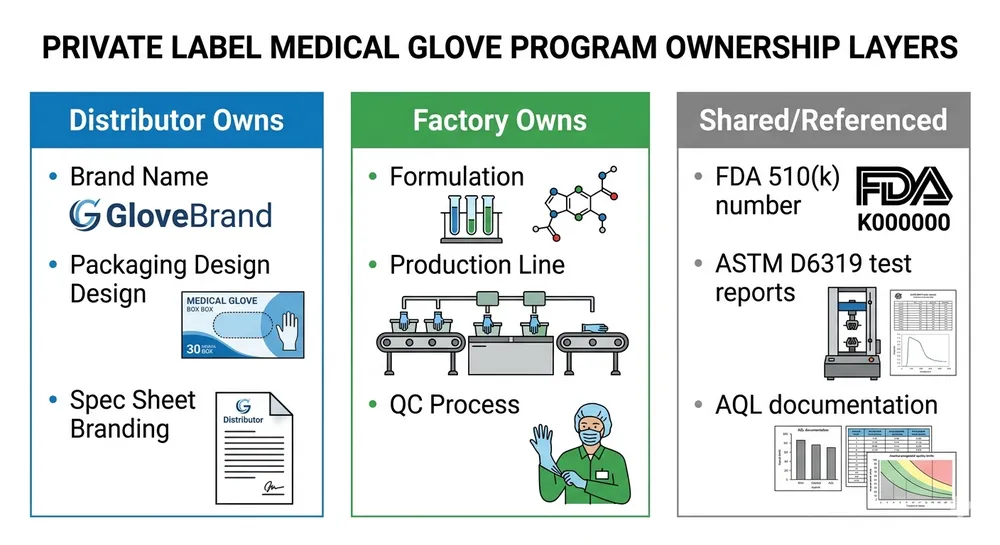
In a private label medical nitrile glove program, you own the brand, the packaging design, and the commercial spec sheet that carries your name. The factory owns the formulation, the production process, and the underlying certifications. You're not buying a formula — you're buying consistent, certified production output that you can brand and resell.
This matters practically because it shapes what documentation you hold and what you reference. Your FDA 510(k) compliance for branded resale doesn't require you to obtain a separate clearance for your label — it requires you to reference the factory's existing 510(k) clearance number in your product documentation and ensure the gloves you're selling were manufactured under that cleared specification. The factory's clearance covers the device; your brand is the commercial layer on top.
What you should lock down in writing before the first container ships:
- Spec sheet under your brand name — thickness range (e.g., 0.08–0.10mm palm, 0.10–0.12mm finger), color, powder-free status, accelerator-free or standard compound, AQL standard, box count (typically 100 or 200 gloves per box)
- Packaging artwork approval — carton label, inner box design, language requirements for US market (English, bilingual if required)
- Certification reference documentation — factory's FDA 510(k) clearance number, ASTM D6319 test report for the specific SKU, ISO 9001 certificate
- AQL documentation — outgoing inspection report per lot, confirming AQL 1.5 or the agreed standard
The spec sheet is the document that protects you. If a container arrives and the gloves don't match the agreed thickness or fail AQL at your warehouse, the spec sheet is what you use to hold the factory accountable. Get it drafted and signed before production starts, not after.
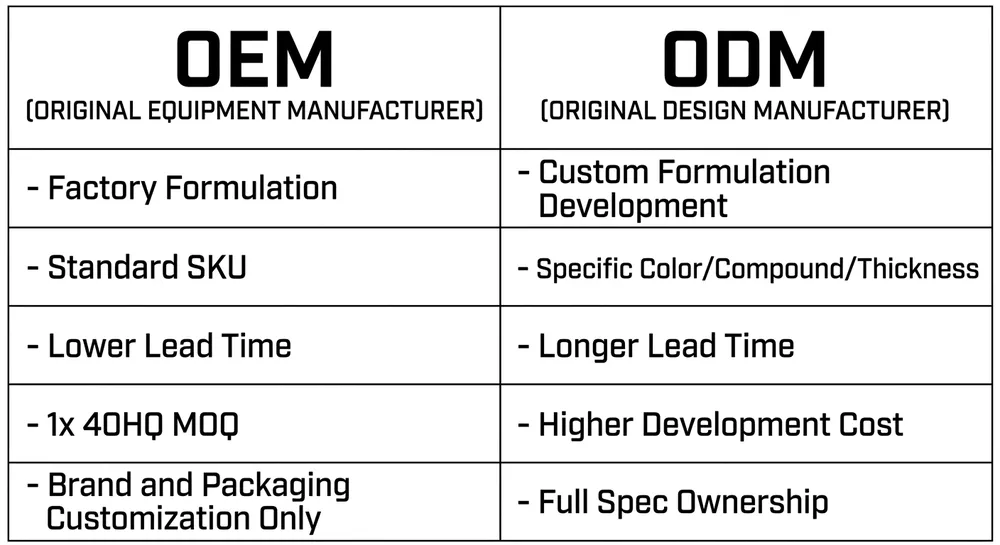
OEM vs ODM: Which Scope Fits Your Program
These two terms get used interchangeably in glove sourcing, but they describe different scopes of work — and the distinction affects your MOQ, your lead time, and how much formulation control you actually have.
OEM (Original Equipment Manufacturer): You're ordering to the factory's existing, validated formulation and spec. The factory has a standard powder-free nitrile examination glove — say, 0.10mm palm thickness, blue, 100-count box — and you're putting your brand on it. The formulation is theirs, the production process is theirs, the certifications are theirs. Your contribution is the brand, the packaging design, and the commercial spec sheet that references their underlying product.
This is the faster, lower-MOQ path. For standard SKUs, MOQ at our facility starts at one 40HQ container — approximately 2,000 boxes for standard examination glove configurations. You're not paying for formulation development or line validation because the product already exists.
ODM (Original Design Manufacturer): You're specifying something that requires formulation or process development — a custom color that isn't in the standard rotation, a specific elongation-at-break target, an accelerator-free compound for a sensitization-sensitive market, or a non-standard thickness profile. The factory develops or adapts the formulation to your spec, validates it, and produces under your brand.
ODM takes longer and typically carries a higher MOQ because the development work has to be amortized across the production run. At our facility, custom formulation ODM projects start at the same one-container minimum, but the timeline includes a formulation development and sample approval phase before production begins. (For accelerator-free compound specifically, we run that on a dedicated line — it's not a special request that requires setting up a new process from scratch, which keeps the lead time reasonable.)
The practical question for most US distributors entering private label: start OEM unless you have a specific market requirement that the factory's standard SKU can't meet. Get a container moving, test it with your customers, and develop a custom formulation in parallel if the market justifies it.
MOQ for Private Label Medical Nitrile Gloves: What the Numbers Actually Mean
MOQ is one of the two questions that stall most private label conversations (compliance is the other). The numbers are straightforward once you understand what drives them.
For standard OEM private label at our facility: one 40HQ container, approximately 2,000 boxes for standard examination glove SKUs (100 gloves per box, 10 boxes per carton). That's roughly 200,000 gloves per container. At that volume, the economics of private label packaging — custom inner box printing, carton labeling, spec sheet production — are absorbed without significantly affecting your per-box cost.
A few things that affect where your actual MOQ lands:
- Size run split: If you're ordering a full size run (XS through XL) in a single container, each size gets a proportionally smaller allocation. We can work with that, but it's worth discussing the split before finalizing the order — some sizes move faster than others, and an unbalanced split can leave you with slow-moving inventory in one size.
- Custom packaging vs. standard: Standard box dimensions and carton configurations are already optimized for 40HQ loading. Custom box sizes can affect carton count per container and therefore your effective MOQ.
- Accelerator-free compound: Same MOQ as standard — one container. We run accelerator-free on a dedicated line, so there's no minimum uplift for the compound itself.
- Custom color: Colors outside the standard rotation (blue, white, black) require a minimum batch for the compound mix. In practice, this usually aligns with the one-container MOQ anyway, but confirm before committing to a color.
Below one container, private label packaging economics don't work — the per-box cost of custom printing on a sub-container run makes the program uncompetitive. If you're testing the market before committing to a full container, the right approach is to order a standard SKU sample (we ship samples within 5–7 business days) and use that to validate demand before the private label investment.
FDA 510(k) and ASTM D6319 Compliance: What US Distributors Actually Need to Hold
This is where a lot of private label programs run into trouble — not because the compliance requirements are complicated, but because distributors don't always know which documents they need to hold versus which ones the factory holds on their behalf.
FDA 510(k) clearance is a device clearance, not a brand clearance. The factory obtains 510(k) clearance for a specific device — in this case, a nitrile examination glove meeting a defined specification. When you sell that glove under your brand, you're selling a device that was manufactured under the factory's cleared specification. You reference the factory's 510(k) clearance number in your product documentation; you don't obtain a separate clearance for your label.
What this means practically: your branded product documentation — spec sheet, product insert, website listing — should reference the factory's FDA 510(k) clearance number. If a US hospital or distributor asks for your FDA documentation, you provide the factory's clearance certificate plus confirmation that your product was manufactured under that cleared specification. The factory should be able to provide a letter of authorization confirming that your branded SKU is covered by their clearance.
(This is the part that trips up first-time private label buyers most often. The FDA clearance question sounds like it requires you to do something — it mostly requires you to document what the factory already has.)
ASTM D6319 is the standard specification for nitrile examination gloves for medical application. It covers physical dimensions, tensile strength, elongation before and after aging, and freedom from holes (AQL). For US market resale, your product documentation should include the factory's ASTM D6319 test report for the specific SKU you're selling — not a generic certificate, but a test report showing the actual measured values for the lot or production run.
The documents you should hold for each private label SKU:
| Document | Who Holds It | What It Covers |
|---|---|---|
| FDA 510(k) clearance certificate | Factory (you reference it) | Device clearance for the glove specification |
| Letter of authorization | Factory issues, you hold | Confirms your branded SKU is covered by their 510(k) |
| ASTM D6319 test report | Factory issues per lot | Physical performance data for the specific production run |
| ISO 9001:2015 certificate | Factory | Quality management system certification |
| AQL 1.5 outgoing inspection report | Factory issues per shipment | Lot-level defect rate documentation |
| CE declaration of conformity | Factory (if selling into EU) | EN 455 compliance for European market |
The AQL documentation is worth a specific note. AQL 1.5 is a tighter standard than the AQL 2.5 that many factories use as their default outgoing inspection. The difference: at AQL 1.5, the acceptable defect rate per lot is lower, which means fewer defective units reach your warehouse. When you're building a private label brand, your downstream customers don't know or care about your factory's QC standard — they just know whether the gloves they received are acceptable. AQL 1.5 is the standard that protects your brand reputation at the distribution level.
For a deeper look at how to verify these certifications before a container ships, see our guide on how to verify medical nitrile glove certifications.

Spec Parameters to Lock Before Your First Container
The spec sheet is a contract. Vague specs produce variable product. These are the parameters worth locking explicitly — not because factories will deliberately deviate, but because "standard" means different things to different production teams.
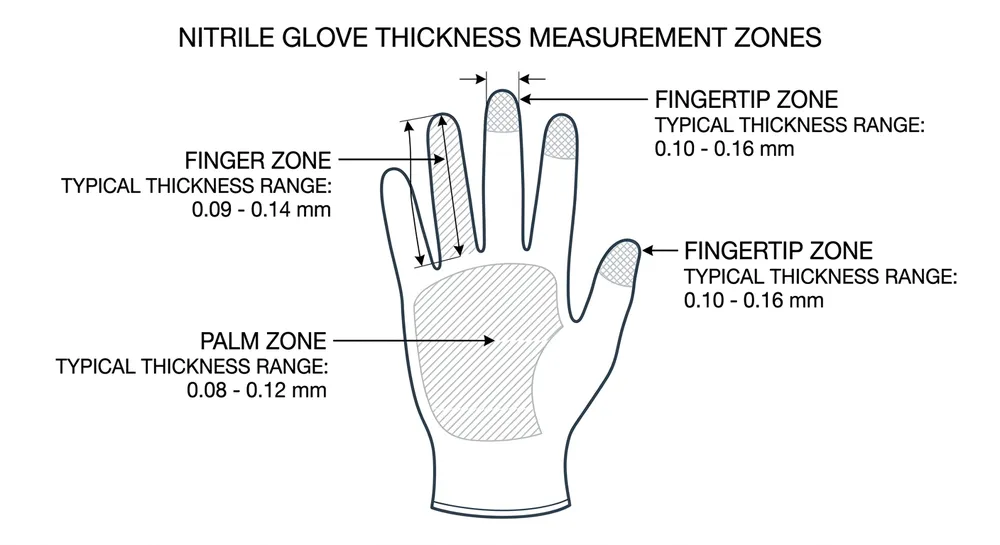
Thickness: Specify palm, finger, and cuff thickness separately, with tolerances. A typical medical examination glove runs 0.08–0.10mm at the palm and 0.10–0.12mm at the finger. Our automated lines hold ±0.01mm tolerance across a production run — but that only matters if the target is written into the spec sheet. "Standard thickness" is not a spec.
Color: Blue, white, and black are standard rotation colors at our facility — no minimum uplift, no development lead time. Custom colors require a Pantone reference and a minimum batch for compound mixing. Confirm before committing.
Compound type: Standard nitrile (accelerator-based vulcanization) or accelerator-free. If you're targeting hospital procurement, dental networks, or any buyer with Type IV allergy sensitivity requirements, accelerator-free is the specification that opens those accounts. Standard compound is fine for general medical, industrial, and food-contact distribution.
Box count: 100 gloves per box is standard for examination gloves. 200-count boxes exist for high-volume clinical settings. The box count affects your per-unit pricing, your carton configuration, and your end-customer's purchasing math — worth specifying explicitly.
Carton labeling format: US market cartons need English-language labeling with lot number, expiry date, and manufacturer information. If you're selling into Amazon FBA or a specific distributor DC, confirm their labeling requirements before artwork approval — some DCs have specific barcode placement and carton dimension requirements that affect how we configure the pack.
Textured fingertip vs. smooth: Textured fingertip improves grip in wet conditions and is standard for most medical examination applications. Smooth finish is available for specific applications. Specify which you need — don't assume.
Red Flags When Evaluating a Private Label Factory
We've had buyers come to us after a failed private label program with another factory. The failure patterns are consistent enough that they're worth naming.
Third-party compounding. If the factory buys pre-mixed nitrile compound from a supplier rather than mixing in-house, they don't control the most important variable in glove consistency. Batch-to-batch variation in the compound translates directly into thickness variation and AQL variance across your orders. Ask specifically: do you mix your own compound? If the answer is vague, that's your answer.
AQL 2.5 as the default standard. Many factories use AQL 2.5 for outgoing inspection. That's an acceptable standard for some applications, but for medical examination gloves going into US healthcare distribution, AQL 1.5 is the standard that keeps your downstream customers from filing complaints. If a factory quotes AQL 2.5 and treats AQL 1.5 as an upgrade tier with a price premium, that tells you something about how they think about quality.
No in-house QC documentation. A factory that can't produce lot-level inspection reports — actual measured values, not just a pass/fail certificate — is a factory that isn't running systematic QC. For private label, you need lot-level documentation because your customers will ask for it, and because it's the only way to trace a quality issue back to a specific production run.
Certifications that don't match the SKU. FDA 510(k) clearance is issued for a specific device specification. If the factory's clearance covers a different thickness range or compound type than what you're ordering, your product isn't covered by that clearance. Ask for the clearance certificate and check that the device description matches your spec. (We've seen factories present a 510(k) certificate for a latex glove program as evidence of their nitrile clearance. It happens.)
No sample approval process. A factory that ships production without a pre-production sample approval step is a factory that doesn't expect to be held to a spec. The sample approval process — where you receive and approve a production sample before the full run begins — is the checkpoint that catches spec drift before it becomes a container problem.
For a full view of our OEM ODM Nitrile Gloves capabilities and how we structure private label programs, that page covers the process in more detail.
Accelerator-Free Private Label: The Specification That Opens Hospital Accounts
Standard nitrile gloves use thiuram and carbamate accelerators in the vulcanization process. These accelerators are the primary cause of Type IV delayed hypersensitivity reactions in healthcare workers — a compliance concern that has moved from niche to mainstream in European hospital procurement over the past several years, and is gaining traction in US healthcare systems as well.
Accelerator-free nitrile uses a sulfur-donor cure system instead. The resulting glove has no detectable accelerator residue and passes EN 455 Part 4 (biological evaluation). For distributors supplying hospital groups, dental networks, or any procurement environment where Type IV allergy documentation is required, accelerator-free is the specification that gets you on the approved vendor list.
We run accelerator-free compound on a dedicated line at our facility — it's not a special project that requires setting up a new process. The MOQ is the same as standard: one 40HQ container. The compound is mixed in-house, which means we control the accelerator-free specification the same way we control our standard formulation — not dependent on a third-party supplier's batch consistency.
If you're building a private label line for US healthcare distribution and you're not sure whether your target accounts require accelerator-free, the practical answer is: ask your top three hospital or clinic accounts whether they have a Type IV allergy policy for glove procurement. If any of them do, accelerator-free is the specification you need. It's easier to launch with the right compound than to requalify a product after your first hospital tender.
Frequently Asked Questions
What is the MOQ for private label medical nitrile gloves?
One 40HQ container — approximately 2,000 boxes for standard examination glove SKUs (100 gloves per box). This applies to both standard OEM programs (factory formulation, your brand) and accelerator-free compound SKUs. Custom formulation ODM projects start at the same container minimum but include a formulation development phase before production.
Do I need my own FDA 510(k) clearance to sell private label nitrile gloves in the US?
No. FDA 510(k) clearance is a device clearance held by the manufacturer. When you sell private label gloves manufactured under the factory's cleared specification, you reference the factory's 510(k) clearance number in your product documentation. The factory should provide a letter of authorization confirming your branded SKU is covered by their clearance. You don't obtain a separate clearance for your label.
What ASTM D6319 documentation should I request from the factory?
Request a lot-level test report — not a generic certificate — showing actual measured values for tensile strength, elongation before and after aging, and AQL hole test results for the specific production run. A certificate that says "meets ASTM D6319" without measured values is not useful documentation for your compliance file or for responding to customer audits.
What's the difference between AQL 1.5 and AQL 2.5 for medical gloves?
AQL (Acceptable Quality Level) defines the maximum acceptable defect rate per lot at a given sampling confidence level. AQL 1.5 is tighter than AQL 2.5 — it means fewer defective units per lot are accepted before the lot is rejected. For medical examination gloves going into US healthcare distribution, AQL 1.5 is the standard that keeps complaint rates low at the distribution level. If a factory quotes AQL 2.5 as their standard, ask whether AQL 1.5 is available and at what cost — the answer tells you how they think about quality.
Can I add accelerator-free nitrile to my private label SKU mix without a higher MOQ?
Yes. At our facility, accelerator-free compound runs on a dedicated line with the same one-container MOQ as standard nitrile. There's no formulation development cost because the compound is already in production rotation. You specify accelerator-free in your spec sheet, and we produce to that spec the same way we produce standard SKUs.
How do I verify that a factory's FDA 510(k) clearance actually covers the gloves I'm ordering?
Request the clearance certificate and check the device description against your spec. The clearance covers a specific device — thickness range, material, intended use. If you're ordering a 0.08–0.10mm powder-free nitrile examination glove and the factory's clearance was issued for a different specification, your product isn't covered. You can also verify 510(k) clearances directly through the FDA's 510(k) database using the clearance number the factory provides.
—
If you're ready to move forward with a private label program, the fastest path is to send us your target spec — glove grade (exam or surgical), size run, target thickness, compound type (standard or accelerator-free), and annual volume estimate. We'll confirm MOQ fit, provide a spec sheet draft under your brand name, and send samples for approval before production begins. Request Quote or browse our full Medical Nitrile Gloves range to see the standard SKUs available for private label.