Medical Nitrile Glove Suppliers for Canadian Healthcare Distributors: Sourcing and Pricing Review
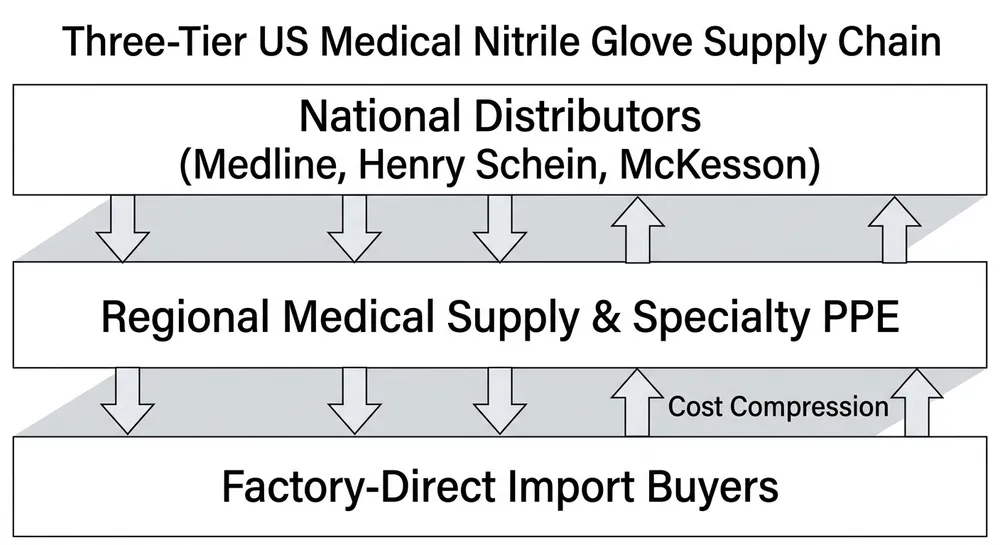
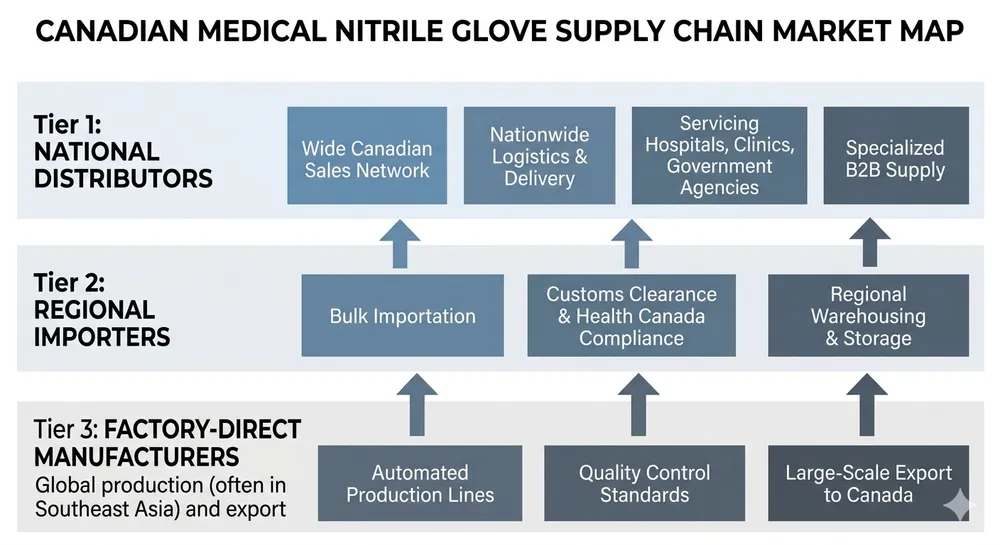
The Canadian medical glove market runs on a small number of large national distributors, a handful of regional players, and an increasingly direct pipeline from overseas manufacturers. If you're sourcing medical nitrile gloves for healthcare distribution in Canada right now, you're navigating all three layers simultaneously — and the pricing gap between them has widened considerably since 2021.
This review covers the main supplier models operating in Canada, what each one is actually good for, and where the economics shift in favor of factory-direct procurement. We've handled pre-shipment documentation and compliance verification for Canadian healthcare distributors for years, so the framing here is practical, not theoretical.
How the Canadian Medical Glove Supply Chain Actually Works
Most Canadian healthcare distributors buy from one of two places: a national distributor who warehouses domestically, or a direct import arrangement with an overseas manufacturer. The middle layer — regional importers who bring in container loads and resell — exists but has thinned out since the pandemic-era supply disruptions reshuffled supplier relationships.
The national distributor model offers real advantages. Stock is on Canadian soil, delivery is measured in days not weeks, and the supplier relationship is straightforward to manage. For urgent replenishment, small top-up orders, or situations where your customer needs product before a regulatory deadline, that local inventory is worth paying for.
The cost of that convenience is real, though. A national distributor is carrying warehousing, logistics, and margin across the chain. By the time medical nitrile gloves reach your invoice, you're typically paying a landed-and-marked-up price that reflects two or three margin layers above the factory gate. For distributors running thin margins on healthcare contracts — especially provincial tender business — that stacked cost structure compresses what you can offer competitively.
The compliance picture in Canada adds another layer. Medical nitrile gloves sold into Canadian healthcare settings need to meet Health Canada's Medical Devices Regulations under the Food and Drugs Act. Class I medical devices (examination gloves) require a Medical Device Establishment Licence (MDEL) for importers and distributors, and the gloves themselves need to meet ASTM D6319 or equivalent performance standards. FDA 510(k) clearance is not automatically recognized in Canada, though it's a useful signal of manufacturing quality. (We see this confusion regularly — buyers assume FDA clearance covers Canadian import requirements. It doesn't, and a container held at the border is an expensive lesson.)
What Separates a Reliable Medical Nitrile Supplier from a Commodity One
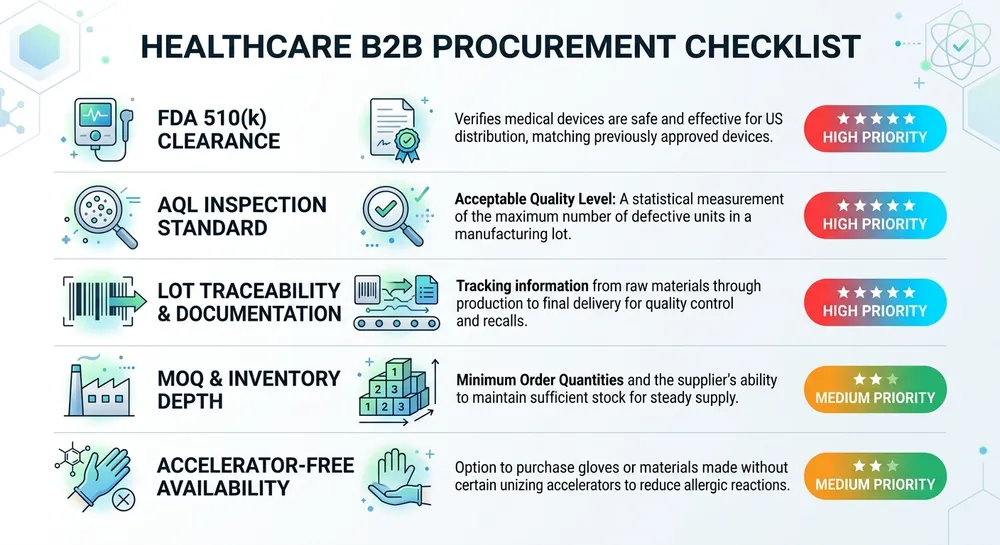
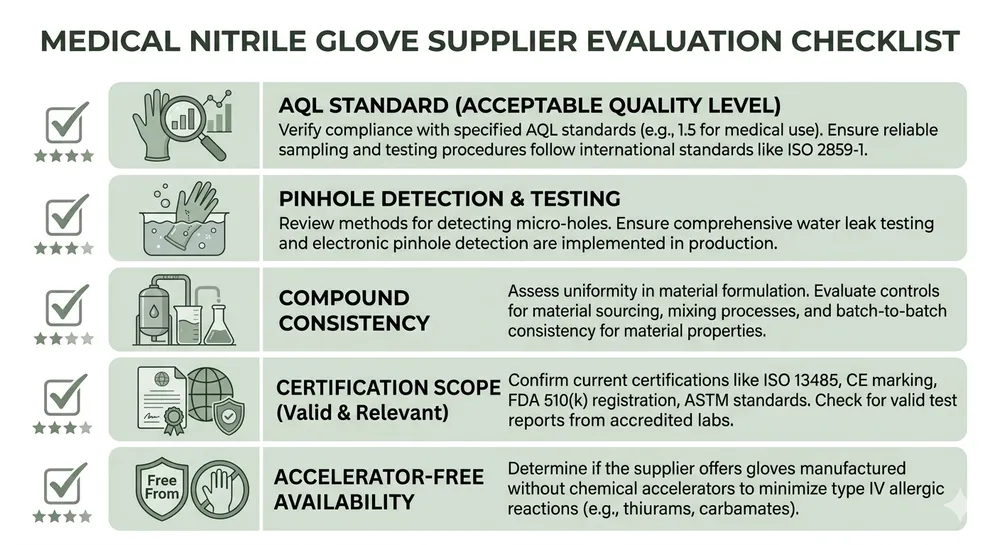
Before walking through the supplier landscape, it's worth being clear about what actually matters when you're evaluating a medical nitrile glove supplier for healthcare distribution. The criteria that protect your business are not the same as the criteria that look good in a product brochure.
AQL standard and inspection methodology — Medical examination gloves should be inspected to AQL 1.5 for critical defects. AQL 2.5 is common in industrial glove supply and is not appropriate for medical-grade product. Ask specifically: is AQL 1.5 applied at the production line, or only at final pre-shipment inspection? The answer tells you a lot about how seriously a manufacturer treats defect control.
Pinhole detection — Online pinhole detection systems running during production are the difference between catching a defect before it ships and discovering it after your customer has opened the box. Not every factory runs them. We run 100% online detection on all eight of our dipping lines — it's not optional for medical-grade output.
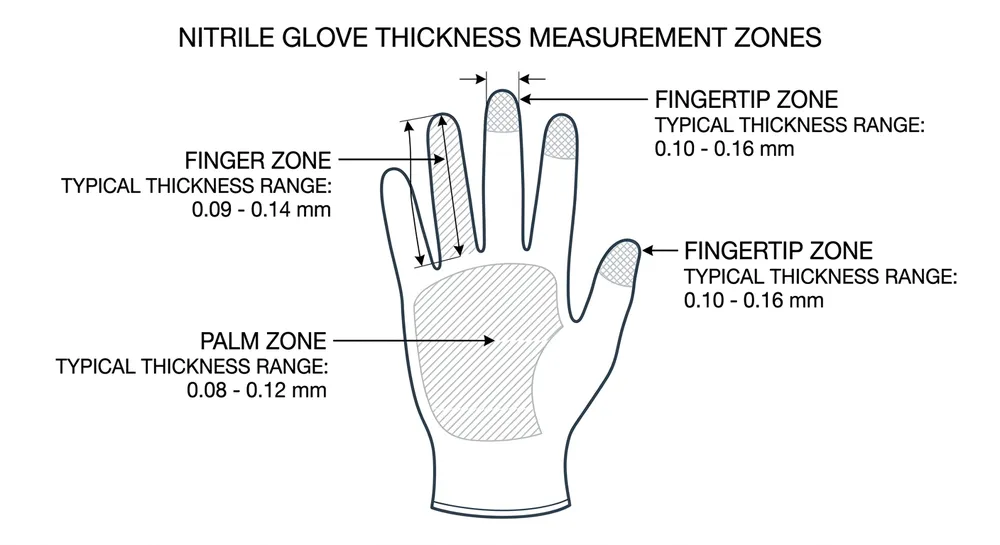
Compound consistency — Nitrile glove performance depends heavily on the formulation. Factories that buy compound from external suppliers have less control over batch-to-batch consistency than factories running their own compounding lab. Thickness variation is the most common downstream complaint we hear from distributors switching suppliers — a glove that measures 0.10mm at the palm and 0.07mm at the fingertip will fail AQL at destination.
Certification scope — ISO 9001:2015 covers quality management systems. EN 455 covers medical glove performance for European markets. ASTM D6319 is the relevant US/Canadian performance standard for nitrile examination gloves. FDA 510(k) clearance means the device has been reviewed for substantial equivalence. A supplier holding all four has been through meaningful external scrutiny. A supplier holding only ISO 9001 has demonstrated process management, not product performance.
Accelerator-free availability — Canadian hospital procurement increasingly specifies accelerator-free nitrile for sensitization-sensitive environments. If your supplier can't offer a Type IV allergy-compliant formulation from the same facility, you're going to lose tenders as that specification becomes standard.
The 10 Suppliers Operating in the Canadian Medical Nitrile Glove Market
The Canadian market is served by a mix of national healthcare distributors, manufacturer-owned distribution arms, and direct-import manufacturers. Here's an honest look at each.
1. McKesson Canada — mckesson.ca
McKesson is the largest healthcare distributor in Canada by revenue and breadth of product range. Their nitrile glove offering spans multiple brands and grades, with national warehouse coverage that makes them the default choice for hospital systems and large clinic networks. The trade-off is that McKesson is a distributor, not a manufacturer — you're buying through their margin layer, and product-level specification flexibility is limited to what they stock. For distributors who need to differentiate on spec or private label, McKesson is not the right channel.
2. Cardinal Health Canada — cardinalhealth.ca
Cardinal Health operates both distribution and manufacturing arms globally, and their Canadian distribution business covers medical consumables including nitrile examination gloves. Their Flexal and similar branded lines are well-established in Canadian hospital procurement. Like McKesson, Cardinal Health's Canadian operation is primarily a distribution business — the manufacturing leverage is in their US and global operations, not locally. Pricing reflects a full distribution margin.
3. Medline Canada — medline.com
Medline is a US-headquartered manufacturer-distributor that operates in Canada. Their model is closer to direct than pure distribution — they manufacture a significant portion of their own product lines — which gives them more pricing flexibility than pure distributors. Their nitrile glove range covers examination and procedure grades. Canadian healthcare distributors buying Medline are getting a more integrated supply chain than with McKesson or Cardinal, though the product range is still standardized rather than customizable.
4. Henry Schein Canada — henryschein.ca
Henry Schein's Canadian operation is strongest in dental and medical office supply. Their nitrile glove offering is broad but oriented toward smaller-volume buyers — dental practices, clinics, and medical offices rather than hospital systems or large-volume distributors. If your customer base is in that segment, Henry Schein is a relevant benchmark for pricing. For high-volume healthcare distribution, their model is less competitive on unit economics.
5. Ansell — ansell.com
Ansell is a global glove manufacturer with strong brand recognition in both medical and industrial segments. Their GAMMEX and MICRO-TOUCH lines are specified in Canadian hospital procurement. Ansell manufactures in Malaysia and other locations and distributes through Canadian healthcare channels. Their medical nitrile range is well-certified and widely accepted. The limitation for distributors is that Ansell sells branded product — private-label or OEM arrangements are not their model, and pricing reflects a premium brand position.
6. Kimberly-Clark Professional — kcprofessional.com
Kimberly-Clark's Purple Nitrile line has significant penetration in Canadian healthcare settings, particularly in surgical and procedure environments. Like Ansell, they sell branded product through distribution channels. Their Canadian presence is strong, but the model is brand-driven rather than specification-flexible. Distributors carrying KC Professional are selling a recognized brand, which has value in certain procurement contexts, but the margin structure reflects that brand premium.
7. Supermax Healthcare — supermaxglove.com
Supermax is a Malaysian nitrile glove manufacturer with North American distribution infrastructure. They supply both branded and private-label product and have been active in the Canadian market, particularly during and after the pandemic supply disruptions. Their manufacturing scale is significant. For distributors looking for a manufacturer-direct relationship with North American distribution support, Supermax is one of the more accessible options. Certification coverage includes FDA 510(k) and EN 455.
8. Sempermed — sempermed.com
Sempermed is an Austrian glove manufacturer (part of the Semperit Group) with distribution into North American healthcare markets including Canada. Their medical nitrile range is well-regarded in European hospital procurement and carries EN 455 certification. Canadian distribution is handled through healthcare supply channels. Sempermed's positioning is quality-focused rather than price-competitive — they're a reasonable benchmark for premium-tier medical nitrile specification.
9. Cranberry USA — cranberryusa.com
Cranberry is a glove manufacturer supplying the North American dental and medical market with a range that includes nitrile examination gloves. They've built distribution into Canadian dental and medical supply channels. Their product range includes standard and specialty formulations. For distributors serving dental and medical office segments, Cranberry is a relevant supplier to benchmark — their pricing tends to be more competitive than the major branded players.
10. Dynarex — dynarex.com
Dynarex is a US-based medical supply company with a broad consumables range including nitrile examination gloves. Their Canadian distribution covers medical and long-term care segments. Dynarex competes on price and breadth of product range rather than premium specification. For distributors supplying cost-sensitive segments — long-term care, home health, community clinics — Dynarex is a relevant pricing reference point.
Where Local Distribution Adds Real Value — and Where It Doesn't
The suppliers above serve the Canadian market well for specific buying situations. Local distribution is genuinely the right answer when:
- You need product in days, not weeks. A hospital system running low on examination gloves mid-contract doesn't have six weeks for a container. Local stock solves that problem, and the premium is worth paying.
- Your order volume is below container minimums. A 200-case top-up order doesn't justify a direct import arrangement. Local distributors exist precisely for this use case.
- You're in a compliance-sensitive procurement cycle. If your customer needs documentation that references a Canadian-registered supplier, local distribution simplifies that paperwork trail.
- You're new to a product category. Starting with a local distributor while you learn the spec requirements and customer preferences is a reasonable risk management approach before committing to container-scale direct import.
These are real advantages. A sourcing review that dismisses them isn't being honest with you.
The economics shift when your volume grows. At repeat container scale — roughly 2,000 boxes per order and above — the cost structure of buying through a national distributor starts to work against your margin. You're paying for warehousing you don't need, logistics you could manage directly, and a margin layer that doesn't add value at your volume. The question isn't whether local distribution is bad. The question is whether it's the right model for your current order profile.
The Landed Cost Gap at Container Scale
The pricing difference between buying through a Canadian distributor and importing direct from a certified manufacturer is not trivial at volume. We're not going to publish a specific number here because it varies by grade, thickness, and market conditions — but the structural gap is consistent.
A national distributor buying from a manufacturer, warehousing in Canada, and selling to you is running at least two margin layers above the factory gate. At commodity nitrile pricing, that gap can represent 20–35% of your unit cost depending on the grade and the distributor's own cost structure. For a distributor running a healthcare contract at fixed pricing, that gap is the difference between a profitable account and a break-even one.
The calculation changes when you factor in the full landed cost of direct import: ocean freight, customs duties (Canada's MFN tariff on nitrile gloves from most manufacturing countries is relatively low), MDEL compliance costs, and the working capital tied up in a container. For buyers doing this for the first time, those costs are real and need to be modeled carefully. For buyers who have run the numbers and are doing repeat container procurement, the economics are usually clear.
(We've helped Canadian distributors model this comparison more times than I can count. The tipping point is almost always around the third or fourth container — by then the process is familiar, the documentation is templated, and the per-unit savings are compounding.)
Factory-Direct as a Structural Sourcing Decision for Medical Nitrile Gloves
Factory-direct import from a certified manufacturer is not a workaround for buyers who can't get local supply. It's a deliberate sourcing decision that makes sense when your volume, margin requirements, and compliance capability align.
At glovemfg, we manufacture medical nitrile gloves across examination and procedure grades from our 18,000 m² facility in Qingdao. Eight fully automated dipping lines run at a combined annual capacity of 1.2 billion pieces. That scale means your container order — typically 2,000 boxes for a standard 20-foot container — doesn't compete with other buyers for line time. We've been running since 2007, and the facility and team are built around consistent output at export quality, not sample-quality production.
The certifications that matter for Canadian healthcare distribution are in place: ISO 9001:2015, CE, EN 455, ASTM D6319, and FDA 510(k). For buyers navigating Health Canada's MDEL requirements, we can provide the technical documentation package — test reports, lot traceability records, and certificate of conformance — that your compliance team needs.
Our in-house compounding lab is the part of our operation that most directly affects what you receive. We formulate our own nitrile compound rather than buying from external suppliers, which gives us control over batch-to-batch consistency that contract manufacturers using third-party compound can't match. The 0.08–0.15mm thickness tolerance we hold across production runs is a function of that compound control combined with our automated dipping line calibration. Thickness variation is the most common quality complaint we hear from distributors who've switched to us from other suppliers — and it's the first thing we address in our pre-shipment AQL 1.5 inspection.
For Canadian distributors who need accelerator-free nitrile — increasingly specified in hospital and long-term care procurement — we produce Type IV allergy-compliant formulations from the same facility. This isn't a separate product line requiring a separate supplier relationship. It's a formulation variant we can run on the same lines with the same QC standards.
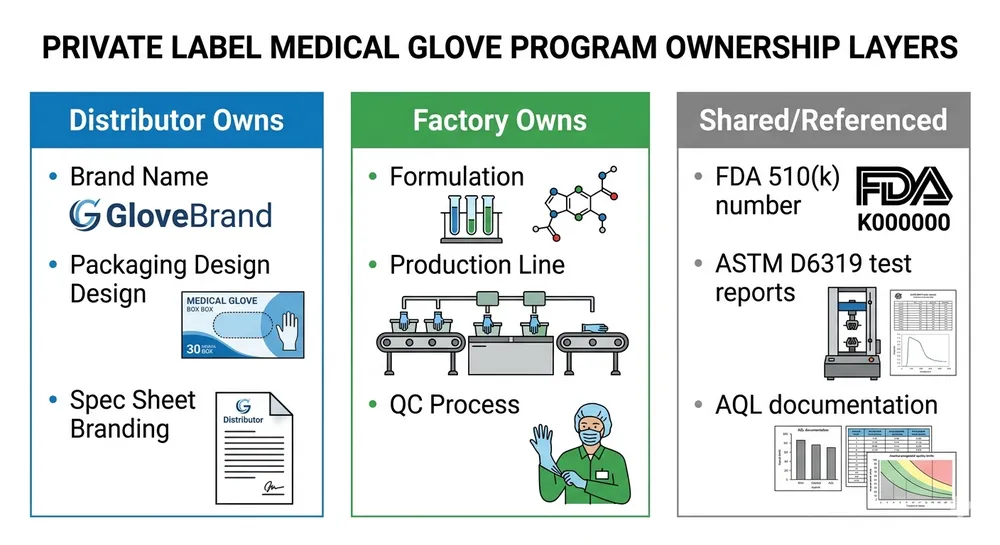
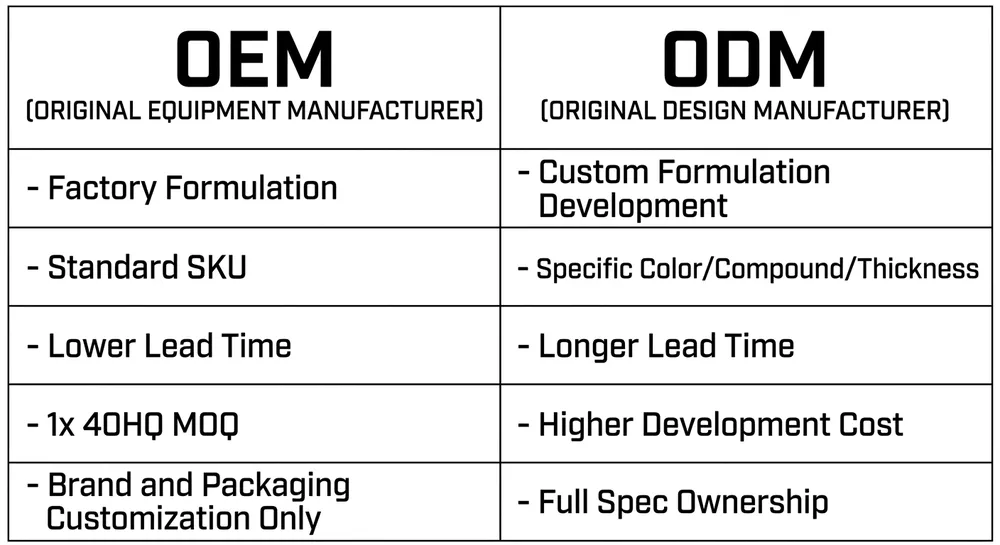
OEM and private-label arrangements are available from a minimum of one container (approximately 2,000 boxes) for standard SKUs. Custom formulations, thickness specifications, and branded packaging are supported with standard lead times. If you're building a private-label medical glove line for the Canadian market, the compliance documentation and OEM specification process is something we've done with Canadian buyers before — it's not a new workflow for us.
Matching Your Sourcing Model to Your Order Profile
The right sourcing decision depends on where you are in your procurement cycle and what your volume looks like. Here's a straightforward framework:
Use local distribution when:
- Order volume is below one container (under ~2,000 boxes)
- Delivery urgency is under two weeks
- You're in a compliance transition and need domestic documentation support
- You're testing a new grade or specification before committing to volume
Consider factory-direct when:
- You're running repeat orders of one container or more
- Your margin on healthcare contracts is under pressure from local pricing
- You need private-label or custom specification that local distributors can't provide
- You're building a product line and need consistent spec across multiple orders
The hybrid model most of our Canadian buyers land on: local distributor for emergency top-ups and small orders, factory-direct for planned procurement cycles. The two models aren't mutually exclusive — they serve different parts of your inventory management problem.
The suppliers listed in this review are real businesses serving the Canadian market. For standard-grade examination gloves at distributor-friendly pricing, McKesson, Cardinal Health, and Medline are the most accessible starting points. For branded product with strong hospital specification acceptance, Ansell and Kimberly-Clark are the benchmarks. For manufacturer-direct relationships with more pricing flexibility, Supermax and Cranberry are worth evaluating alongside a factory-direct option.
What none of the distributor-model suppliers can offer is the combination of custom formulation, private-label OEM, and direct factory pricing at container scale. That's where the economics of a direct manufacturing relationship become relevant to your business.
Frequently Asked Questions
What certifications do medical nitrile gloves need for Canadian healthcare distribution?
Gloves sold as medical devices in Canada must comply with Health Canada's Medical Devices Regulations. Importers and distributors need a Medical Device Establishment Licence (MDEL). The gloves themselves should meet ASTM D6319 (the standard for nitrile examination gloves) or equivalent performance standards. FDA 510(k) clearance is a useful quality signal but does not substitute for Canadian regulatory compliance. EN 455 certification, while a European standard, is widely accepted as evidence of medical-grade performance in Canadian procurement.
What is AQL 1.5 and why does it matter for medical gloves?
AQL (Acceptable Quality Level) 1.5 means that in a statistically sampled inspection, no more than 1.5% of units in a lot may contain critical defects. For medical examination gloves, AQL 1.5 is the appropriate standard — it's more stringent than AQL 2.5, which is common in industrial glove supply. When evaluating a supplier, ask whether AQL 1.5 is applied at the production line or only at final pre-shipment inspection. Production-line inspection catches defects before they compound across a batch.
What is the minimum order for factory-direct medical nitrile gloves from China?
For standard SKUs, one 20-foot container — approximately 2,000 boxes of 100 gloves — is a workable minimum for most manufacturers including glovemfg. Below that volume, the logistics economics don't favor direct import. For OEM or custom-specification orders, MOQ may be higher depending on the formulation and packaging requirements. Most Canadian distributors doing factory-direct procurement run at one to three containers per order cycle.
Are accelerator-free nitrile gloves available from factory-direct suppliers?
Yes. Accelerator-free nitrile — formulated without thiurams, carbamates, and mercaptobenzothiazole accelerators — is available from manufacturers running their own compounding labs. It's not a separate product category requiring a different supplier; it's a formulation variant. For Canadian hospital and long-term care procurement where Type IV allergy sensitivity is a specification requirement, confirm that your supplier can provide the relevant test documentation demonstrating accelerator-free compound, not just a label claim.
How do I verify a medical nitrile glove supplier's certifications before placing a container order?
Request the actual certificate documents, not just a list of certifications. For FDA 510(k), the clearance number is publicly searchable in the FDA's 510(k) database — verify it matches the specific product and manufacturer. For EN 455, the certificate should name the notified body that issued it and include an expiry date. For ISO 9001:2015, the certificate should name the certification body and the scope of certification. Pre-shipment, request the lot-specific test report and certificate of conformance for your order — these should reference the specific production lot, not just the product line.
—
If your current sourcing model is under margin pressure or you're evaluating a private-label medical nitrile line for the Canadian market, the comparison between local distribution and factory-direct procurement is worth running with real numbers. We can provide a landed-cost comparison for your specific grade, volume, and delivery requirements — Request Quote with your target specification and we'll put together a detailed breakdown.
For more on our medical nitrile glove range and available grades, visit our Medical Nitrile Gloves category page.
“`