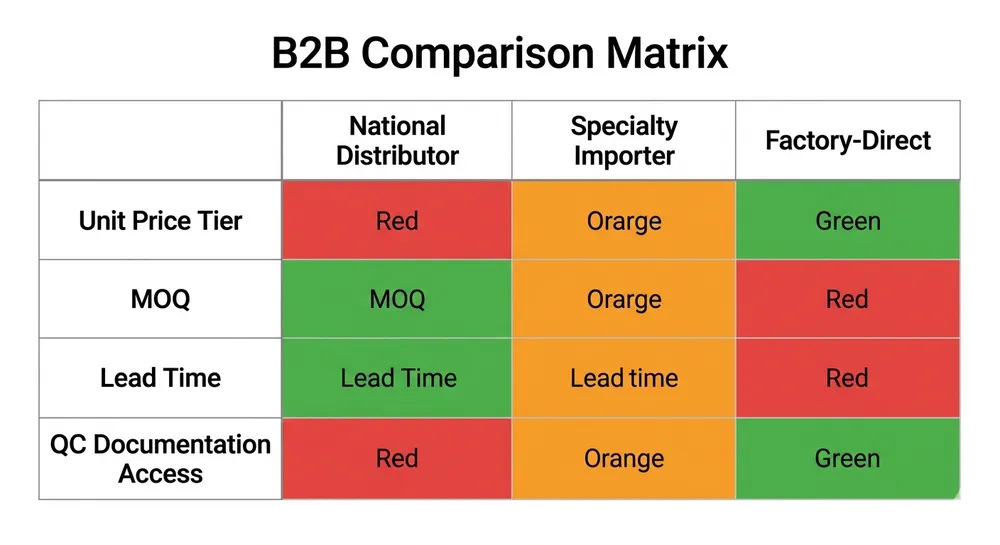
Vinyl is cheaper per box. That's the whole argument for it — and for some buyers, it's enough. But "cheaper per box" is not the same as "lower cost per deployment," and when you're moving containers, the gap between those two numbers can be significant.
Here's the short verdict: nitrile wins on protection, compliance coverage, and total cost of ownership for most commercial applications. Vinyl wins on unit price for low-risk, short-contact tasks where regulatory requirements are minimal. If your buyers are in food service, light janitorial, or non-medical general handling, vinyl can work. If they're in healthcare, industrial, or any regulated environment, vinyl creates compliance exposure that nitrile doesn't.
The rest of this article shows you exactly where that line sits — and what it costs you when you're on the wrong side of it.
—
Head-to-Head: What the Spec Difference Actually Means at the Port
Before the commercial analysis, the material properties — because they drive everything downstream.
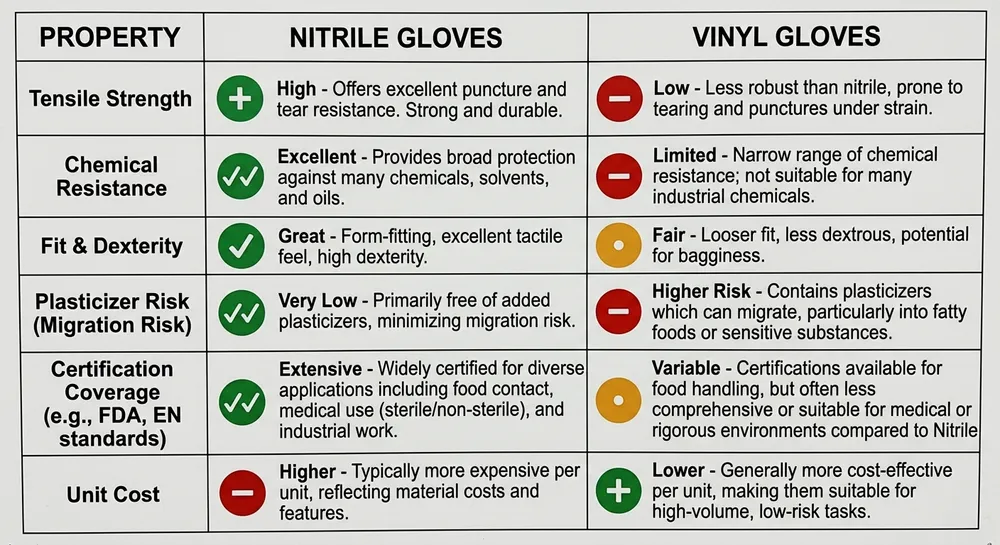
| Property | Nitrile | Vinyl |
|---|---|---|
| Base material | Synthetic rubber (acrylonitrile-butadiene) | PVC with plasticizer |
| Typical thickness | 0.08–0.12mm (medical); 0.10–0.15mm (industrial) | 0.07–0.10mm |
| Tensile strength | High — resists puncture and tearing under stress | Lower — tears more readily under lateral stress |
| Chemical resistance | Broad: oils, solvents, many acids | Limited: water-based solutions only |
| Fit and dexterity | Conforms to hand shape with use | Loose fit, limited tactile feedback |
| Latex protein risk | None | None |
| Plasticizer migration | None | Present — DEHP/DOP concerns in food and medical use |
| Key certifications available | FDA 510(k), ASTM D6319, EN 455, CE | ASTM D5250, limited medical-grade options |
| Relative unit cost | Higher | Lower (typically 20–40% less per box at volume) |
The plasticizer migration row is the one most buyers miss on first pass. Vinyl gloves contain plasticizers — typically DEHP or DOP — to keep the PVC flexible. In food contact and medical applications, plasticizer migration is a regulatory concern in the EU and increasingly scrutinized in US FDA guidance. If your buyers are in those segments, vinyl's cost advantage disappears the moment a compliance question surfaces.
(We see this regularly with European distributors — they'll trial vinyl for cost savings, then pull back when their food-industry customers flag the plasticizer issue during their own supplier audits.)
—
The Hidden Cost: Where Vinyl's Price Advantage Erodes
The 20–40% unit price gap looks compelling in a spreadsheet. Here's what it doesn't show.
Rejection and compliance exposure. Vinyl gloves sold into medical or regulated food-contact channels without proper documentation create liability for the distributor, not just the end user. A single customs query or downstream compliance audit can freeze a shipment or trigger a product recall. The cost of one rejected container exceeds the savings from a year of lower unit pricing.
Failure rate under real use conditions. Vinyl's lower tensile strength means higher in-use failure rates in anything beyond light-contact tasks. In healthcare settings, a glove that tears during a procedure isn't just a product failure — it's an incident report. Your buyer absorbs that cost in staff time, replacement product, and reputational risk with their own customers.
Fit and donning waste. Vinyl's loose fit means more gloves used per task in high-turnover environments. Workers double-glove more often, discard gloves that slip, or use two gloves where one properly fitted nitrile glove would suffice. At 1,000+ employees in a food processing facility, that waste adds up to real per-unit cost that never appears in the purchase order comparison.
Certification documentation gaps. If your buyers need to provide compliance documentation to their own customers — hospitals, government contracts, food manufacturers — vinyl's limited certification coverage creates a documentation gap. Nitrile with FDA 510(k) and ASTM D6319 covers most regulated procurement requirements in North America. Vinyl typically cannot.
We've had buyers come to us after switching to vinyl for a full container cycle, then switching back after their downstream customers started asking for AQL test reports and FDA registration numbers that their vinyl supplier couldn't provide. The switching cost — requalification, new samples, updated documentation — ate the savings from two container orders.
—
Application Showdown: Which Option Wins Where
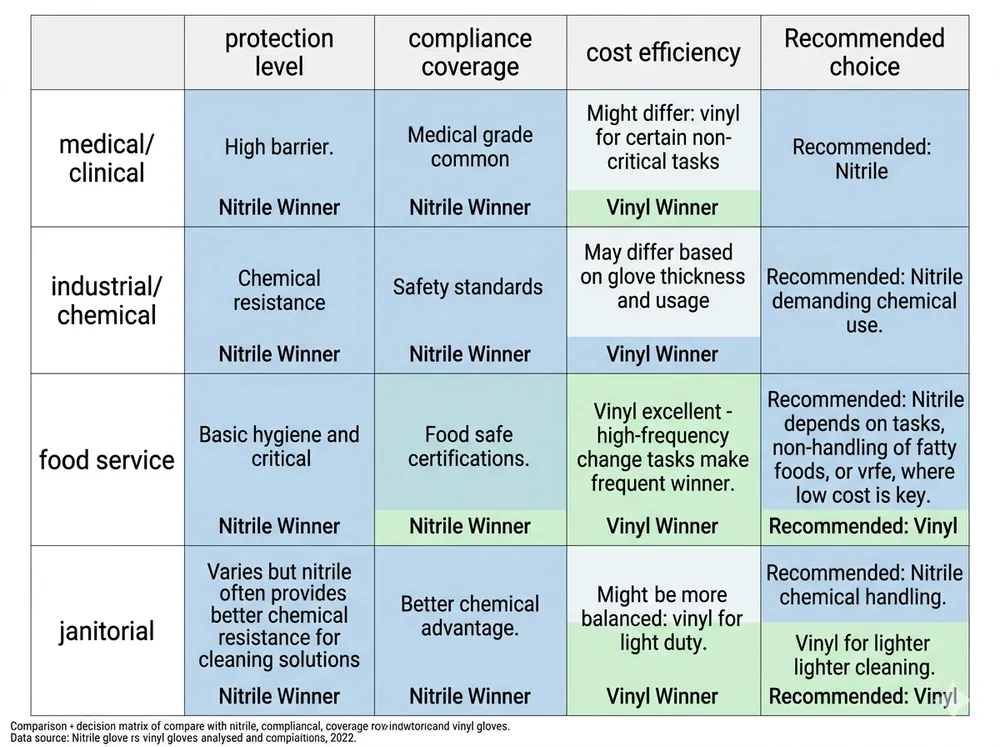
Scenario 1: Medical and Clinical Supply Distribution
Winner: Nitrile — no contest.
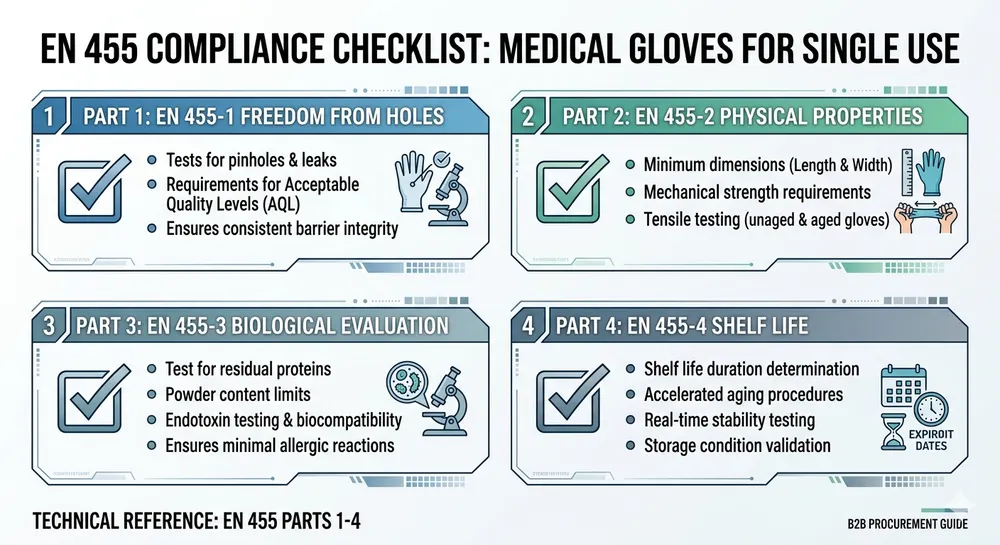
Medical examination gloves require FDA 510(k) clearance in the US and EN 455 compliance in Europe. Vinyl medical gloves exist but represent a shrinking share of the market precisely because their performance profile doesn't meet the expectations of clinical buyers. If your distribution channel touches hospitals, clinics, dental offices, or any regulated healthcare setting, nitrile is the only commercially viable option. Sourcing vinyl here isn't a cost decision — it's a compliance risk.
Scenario 2: Industrial and Chemical Handling
Winner: Nitrile.
Chemical resistance is the deciding factor. Vinyl offers minimal protection against oils, solvents, and most industrial chemicals. Nitrile's acrylonitrile content provides meaningful resistance across a broad chemical spectrum. For automotive, manufacturing, laboratory, and maintenance applications, vinyl fails the basic protection requirement. Your industrial buyers will reject it on spec before price is even discussed.
Scenario 3: Food Service and Light General Handling
Winner: Vinyl on price, nitrile on compliance trajectory.
This is the one scenario where vinyl genuinely competes. For short-contact food handling tasks — sandwich prep, deli counter, light kitchen work — vinyl's protection level is adequate and its lower cost is a real advantage. Many food service operators run vinyl for exactly this reason.
The caveat: EU food contact regulations and evolving FDA guidance on plasticizer migration are tightening. If your food service buyers operate in or export to European markets, or if they supply to food manufacturers with their own supplier compliance programs, vinyl's regulatory position is weakening. Nitrile powder-free is the safer long-term specification for food contact, and the price gap has narrowed enough that many buyers are making the switch proactively.
(This segment has shifted noticeably in the last three years — we've seen a steady migration from vinyl to nitrile among food service distributors who supply both retail and institutional channels.)
Scenario 4: High-Volume Janitorial and Facilities Management
Winner: Depends on task profile.
Janitorial is genuinely split. Light cleaning, trash handling, and surface wiping — vinyl works. Chemical cleaning agents, disinfectants, and any task involving prolonged contact with cleaning solutions — nitrile. The practical answer for most facilities management distributors is to carry both: vinyl for light-duty SKUs, nitrile for chemical-contact SKUs. The margin on nitrile is better, and the upsell from vinyl to nitrile is an easy conversation when you can show the chemical resistance data.
—
What to Verify Before You Source Either Option
This is where procurement decisions go wrong — not in the spec comparison, but in the supplier validation step.
For nitrile, verify:
- FDA 510(k) registration number — not just a certificate copy. Look up the registration number directly in the FDA 510(k) database. We've seen third-party certificates that reference registration numbers belonging to different products or manufacturers.
- ASTM D6319 test reports — request the actual test data, not just a compliance statement. Key parameters: tensile strength before and after aging, elongation at break, AQL 1.5 pinhole test results.
- Thickness consistency across the glove — palm thickness and fingertip thickness should both be specified. A glove that meets palm thickness spec but thins out at the fingertip fails AQL at the destination port. Ask for the tolerance range, not just the nominal spec.
- Powder-free verification — residual powder content should be tested, not assumed. Our production runs 100% powder-free with online verification at the dipping line.
For vinyl, verify:
- Plasticizer type and content — if your buyers operate in EU food contact or medical channels, request DEHP/DOP content documentation. Some vinyl manufacturers have moved to alternative plasticizers; most haven't.
- ASTM D5250 compliance — the standard for vinyl examination gloves. Not all vinyl gloves on the market are tested to this standard.
- AQL level — vinyl is frequently sold at AQL 2.5 or 4.0. If your buyers need AQL 1.5, confirm it explicitly. The difference in defect rate is commercially meaningful at container volumes.
RFQ language that protects you:
When requesting quotes for either material, specify: grade (medical/industrial/food contact), thickness range (nominal and tolerance), AQL level, certification requirements by market, and whether accelerator-free formulation is required. Vague RFQs produce vague quotes — and the gaps show up at the port or in your buyer's warehouse.
—
Cost-Performance Positioning: Where Each Option Sits

The cost-performance gap between nitrile and vinyl has narrowed over the past several years as nitrile raw material costs have stabilized and production volumes have scaled. The 20–40% price premium for nitrile that existed five years ago is closer to 15–25% today for standard powder-free medical grade at container volumes — and that gap continues to compress.
For buyers building a product line, the margin story on nitrile is also better. Nitrile commands a higher retail and institutional price point, and the compliance documentation that comes with a properly certified nitrile product (FDA 510(k), ASTM D6319, AQL 1.5 test reports) is a genuine differentiator your buyers can use to win regulated accounts. Vinyl doesn't give you that sales tool.
Our 8 automated dipping lines run exclusively nitrile — we don't produce vinyl, and we made that decision deliberately. Nitrile's formulation complexity is higher, but the consistency you get from a dedicated facility running one compound type is worth it. Thickness tolerance across our production runs 0.08–0.15mm with online pinhole detection at every line, so the AQL 1.5 standard isn't aspirational — it's what the line produces.
—
Frequently Asked Questions
Are vinyl gloves safe for food contact?
In most markets, yes — for light, short-contact food handling tasks. The concern is plasticizer migration (DEHP/DOP) in prolonged food contact applications, which is regulated under EU food contact materials legislation and increasingly scrutinized in FDA guidance. For food manufacturers or processors with their own supplier compliance programs, nitrile powder-free is the safer specification. For basic food service use, vinyl remains widely accepted.
Can vinyl gloves be used in medical settings?
Vinyl examination gloves exist and are FDA-cleared under ASTM D5250, but their use in clinical settings has declined significantly. Most hospital procurement standards now specify nitrile due to its superior tensile strength, lower failure rate, and broader chemical resistance. If your healthcare buyers have specific procurement standards, check whether vinyl is still on their approved list — many have removed it.
What AQL level should I specify for high-volume procurement?
AQL 1.5 is the standard for medical-grade disposable gloves and should be your baseline for any regulated application. AQL 2.5 is common in industrial and general-use gloves. At container volumes, the difference in defect rate between AQL 1.5 and AQL 2.5 translates to a meaningful number of non-conforming units — specify it explicitly in your RFQ rather than assuming.
Is accelerator-free nitrile worth the premium?
For buyers supplying healthcare or any segment with latex-sensitive or chemical-sensitive end users, yes. Accelerator-free (Type IV allergy-safe) nitrile eliminates the thiuram and carbamate accelerators that cause delayed hypersensitivity reactions. It commands a price premium and is increasingly specified in European healthcare procurement. We produce accelerator-free variants from the same facility — it's a formulation change at the compounding stage, not a different production line.
What's the minimum order for a mixed nitrile/vinyl trial?
We only manufacture nitrile. For a Disposable Nitrile Gloves trial order, our standard MOQ is approximately 2,000 boxes (one container) for standard powder-free medical SKUs. OEM and custom specifications are supported from the same MOQ. If you're evaluating a switch from vinyl to nitrile, we can provide pre-shipment samples with full test documentation before you commit to a container.
—
The Sourcing Decision in Plain Terms
Vinyl has one real advantage: unit price. Everything else — protection, compliance coverage, chemical resistance, fit, certification documentation, and long-term regulatory trajectory — favors nitrile.
The buyers who stay on vinyl long-term are typically in low-risk, non-regulated applications where the compliance question never comes up. The buyers who switch to nitrile usually do it after one of three events: a compliance audit, a downstream customer requirement, or a product failure incident. Switching after one of those events costs more than switching proactively.
If you're building a distribution business in healthcare, industrial safety, or food manufacturing supply, nitrile is the right foundation. If you're supplying light general-use markets and price is the primary competitive lever, vinyl can work — but go in with clear eyes on where the regulatory floor is moving.
For volume pricing, grade selection, and compliance documentation on Disposable Nitrile Gloves, send your requirements to our team via Request Quote — grade, target market, annual volume, and any certification requirements your buyers specify. We'll come back with pricing and the relevant test documentation for your market.